TL;DR: Scientists mapped a ctenophore’s sensory integration center in stunning 3D detail, revealing 17 distinct cell types and a blueprint for how nervous systems first evolved to process multiple senses at once.
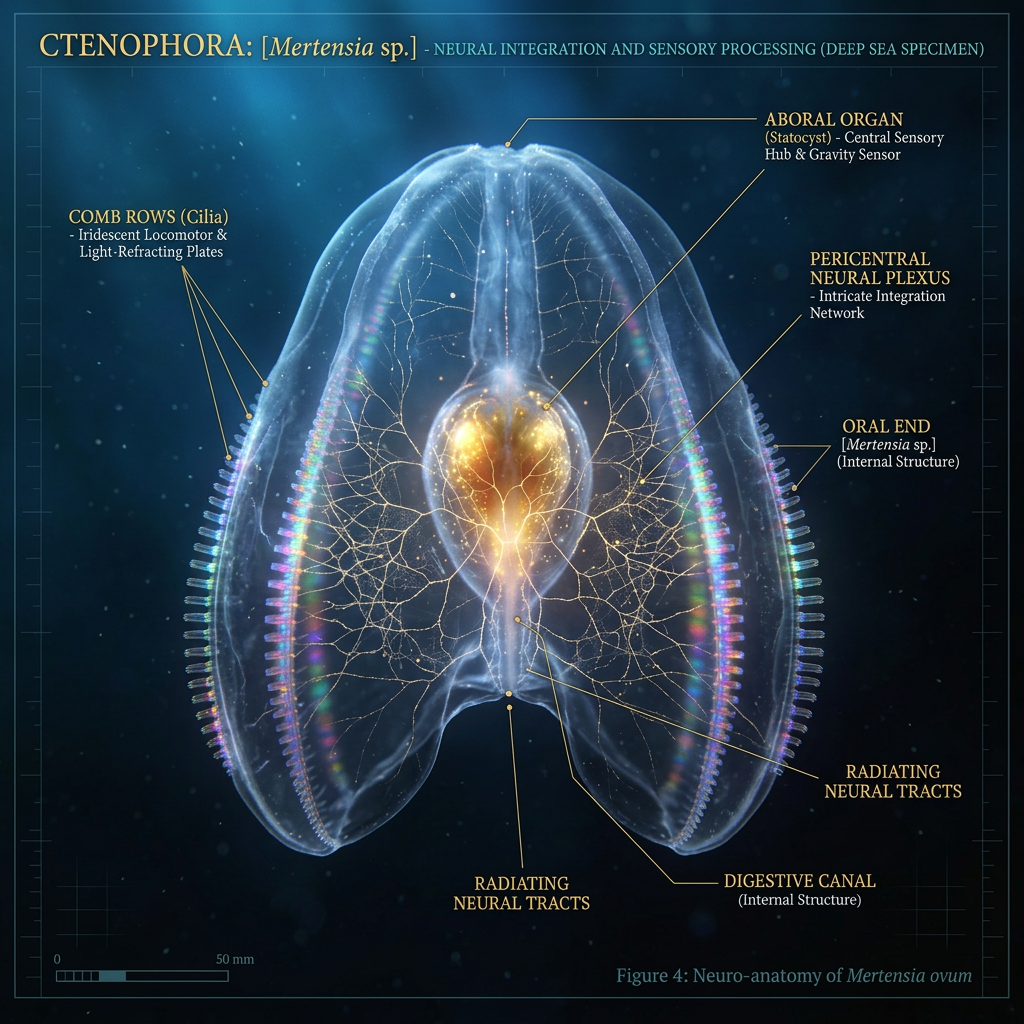
The ctenophore (pronounced “tee-noh-for”), or comb jelly, is a see-through marine animal barely bigger than your pinky. It has no brain—just a simple nerve net. Yet somehow it hunts, avoids predators, and navigates the ocean with remarkable precision. A new study in Science Advances reveals the secret: a microscopic sensory hub called the aboral organ that processes light, motion, and gravity in a way that foreshadows how brains evolved. Using electron microscopy technology so advanced it can trace individual nerve connections in 3D, researchers from Oxford and other institutions mapped this organ cell by cell—and what they found challenges what we thought we knew about the origins of the nervous system.
Key Findings
- 17 cell types identified: The aboral organ contains approximately 900 cells organized into 17 structurally and functionally distinct types—more neural diversity than expected in a creature with no brain.
- Dual-signal transmission system: Cells communicate via both synaptic connections (the classical brain mechanism) and nonsynaptic transmission through volume release of neurotransmitters, suggesting an ancient pathway predating complex brains.
- Integrated sensory processing: The organ contains specialized receptor types for light (photoreceptors), gravity (gravitoreceptors), and motion (mechanoreceptors), all wired into a condensed nerve net that coordinates behavior in real time.
- Gene expression reveals homology: Genetic analysis shows the aboral organ expresses transcription factors associated with apical organs across other animal groups, suggesting a conserved evolutionary origin.
- Multimodal circuit architecture: The organ uses at least two parallel neural circuits—one conventional and one “condensed”—to process different sensory streams simultaneously without a centralized brain.
- Evolutionary marker: The presence of an organized, integrative sensory organ in ctenophores (which branched off from other animals 560 million years ago) implies that sensory integration—a core function of all brains—evolved earlier and more modularly than previously thought.
Source: Science Advances (2026) | Ferraioli et al.
A Microscopic Marvel Hidden in Plain Sight
The aboral organ sits at the apex of the ctenophore’s bell-shaped body, roughly where a brain would be if the animal had one. Despite its position and function, it’s not a true brain—it’s a sensory ganglion, a clustering of nerve cells devoted to detecting and responding to the environment. For years, scientists understood it could sense light, gravity, and motion, but the cellular hardware remained a mystery.
That changed when researchers applied volume electron microscopy (vEM)—a technique that slices tissue into thousands of ultrathin sections and photographs each one. By stitching these images together computationally, scientists can reconstruct neural tissue in 3D with nanometer precision, revealing every synapse and neurotransmitter vesicle.
A Census of Neural Diversity
The reconstruction revealed an unexpectedly complex architecture. The 900-cell organ is not a homogeneous blob but a carefully organized structure composed of eight balancer cells, multiple types of sensory receptors, a network of interneurons, and secretory cells that release neurotransmitters into the tissue space.
Each cell type appears specialized. Balancer cells—the organ’s signature component—carry long cilia and contain sensor-like structures for detecting motion and gravity. Photoreceptors at the organ’s core absorb light. Interneurons integrate signals from sensory cells and relay them toward motor output. Secretory cells create chemical gradients that influence the entire system, suggesting the organ operates as an integrated sensory processor, not just a collection of independent sensors.
Two Routes for Signal Flow
The most striking discovery was how cells communicate. Researchers expected classical synaptic connections—the tiny gaps where neurons release neurotransmitters and receptors catch them. They found plenty of those. But they also discovered extensive nonsynaptic communication: cells releasing neurotransmitters directly into the surrounding tissue fluid, where they wash over nearby cells like a chemical bath.
This dual-channel system is not inefficient—it’s elegant. Synaptic transmission is fast and precise, allowing neurons to send specific “messages” over defined pathways. Nonsynaptic transmission is slower but broadcasts to many cells at once, useful for coordinating widespread responses. The ctenophore’s aboral organ uses both, suggesting that early nervous systems relied more heavily on volume transmission before synaptic networks became dominant.

A Gateway to Evolutionary Origins
Ctenophores are among the earliest-branching animals alive today. They split from the common ancestor of all other animals roughly 560 million years ago, before the explosion of complex nervous systems during the Cambrian period. That ancient lineage makes them a window into what the earliest neural circuits looked like.
The genetic signature of the aboral organ strongly suggests it evolved from the same ancestral structure that gave rise to apical organs in other animal groups. Researchers found that the organ expresses key transcription factors—FoxQ2, Pax6, Mirr—that control sensory organ development across cnidarians (jellyfish, sea anemones), bilaterians (all other animals), and even sponges. This genetic overlap indicates a deep evolutionary homology: the genes that build sensory integration centers are ancient, preserved across hundreds of millions of years.
Rethinking Nervous System Evolution
The standard narrative has long been that nervous systems evolved incrementally: first, simple nerve nets (as in jellyfish and hydra), then nerve cords (in worms), then ganglia, then centralized brains. The ctenophore complicates this story. Its aboral organ is not a primitive nerve net—it’s a genuine integrative center, capable of binding multiple sensory streams into coordinated behavior, and it sits on a branch of the animal tree that diverged before most nervous systems we study evolved.
This suggests sensory integration—the fundamental job of all brains—emerged earlier and more independently than expected. Instead of a single linear path from diffuse to centralized, nervous systems may have evolved through multiple routes, some emphasizing modular integration in specialized ganglia rather than centralized control.
Implications and Open Questions
Understanding how the aboral organ works offers clues about the molecular toolbox nervous systems use. The prominence of nonsynaptic transmission, the diversity of cell types in a small space, and the parallel processing circuits all point to organizational principles that predate and underlie more complex brains.
The researchers also identified previously unknown cell types and synaptic connection patterns, some not yet seen in other organisms. The presence of specialized secretory cells and electron-dense vesicles suggests chemical signaling is far more sophisticated than a simple diffusion model would predict. These discoveries may reshape how neuroscientists think about signal processing in even advanced nervous systems.
The work opens new avenues for studying how sensory systems became increasingly integrated over evolutionary time, and why brains took on the architecture they did. If early nervous systems relied heavily on volume transmission and modular integration, understanding those principles could inform artificial neural networks designed to process multiple sensory streams in compact, efficient ways.
Citation: Ferraioli A, Digel L, Sturm D, Colgren J, Le Goff C, Jan A, Soto-Angel J, Naumann B, Kittelmann M, Burkhardt P. The 3D architecture of the ctenophore aboral organ and the evolution of complex integrative centers in animals. Science Advances. 2026;12(10):eaea8399. DOI: 10.1126/sciadv.aea8399
Authors’ affiliations: Michael Sars Centre, University of Bergen, Norway; University of Rostock, Germany; Oxford University, UK; and other institutions across Europe and North America.