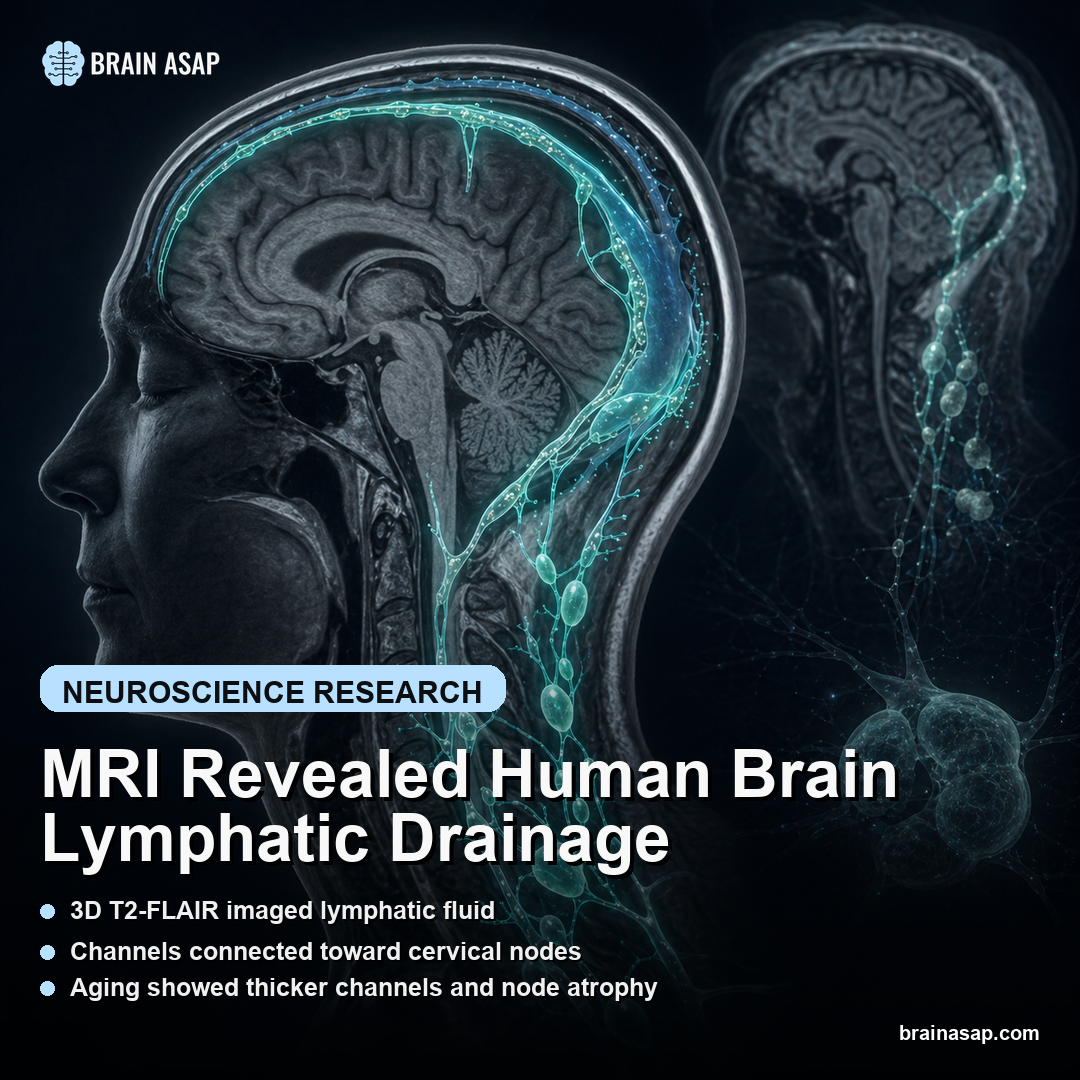
TL;DR: A 2022 study in Nature Communications used non-contrast 3D T2-FLAIR MRI, a scan that suppresses ordinary cerebrospinal-fluid brightness, to map possible brain-border drainage routes toward cervical lymph nodes, but FLAIR brightness alone does not prove lymph flow.
Key Findings
- 81 clinical MRI scans supplied the human map: The retrospective cohort included 45 females and 36 males, ages 15 to 80, scanned on a 3-T Siemens Prisma system without contrast injection.
- Dorsal and ventral routes both appeared: Hyperintense structures were reported along dural venous sinuses, cranial nerve foramina, jugular pathways, and regions leading toward cervical lymph nodes.
- All ventral elements were visible in all subjects: Researchers reported consistent ventral readouts, while some dorsal posterior and jugular regions appeared only in subsets of participants.
- Age over 50 shifted the drainage readout: Older participants had lower dorsal and ventral readout-to-thickness ratios, higher cerebrospinal fluid (CSF), the fluid surrounding the brain and spinal cord volume, thicker channels, and smaller cervical lymph-node measurements.
- The MRI readout is scientifically disputed: A later critique argued that FLAIR hyperintensity can reflect artifacts, CSF flow, venous structures, arachnoid granulations, or protein-rich fluid rather than lymphatic vessels themselves.
Source: Nature Communications (2022) | Albayram et al.
Brain lymphatics are a strange anatomical problem: the brain produces waste like every other organ, yet for decades it seemed to lack the ordinary plumbing that carries fluid, proteins, and immune traffic out of tissues.
This Nature Communications paper asked whether a non-contrast MRI sequence can make part of that plumbing visible in living people, not just in animal dissections or postmortem tissue.
A No-Dye MRI Readout Turned Waste Clearance Into an Imaging Question
The central trick was not a new lymphatic tracer.
It was a familiar MRI contrast problem turned toward a hard anatomical target.
Researchers used 3D T2-FLAIR, a sequence designed to suppress ordinary cerebrospinal fluid while leaving certain protein-rich or macromolecule-rich spaces bright.
Previous human work often depended on gadolinium contrast, delayed imaging, autopsy, or indirect flow evidence. A non-contrast approach would be easier to repeat in aging cohorts and disease studies.
The team first used albumin phantoms to ask whether the sequence can brighten protein-rich fluid while keeping low-protein CSF dark. In their clinical images, they then looked for bright linear structures in anatomical regions where animal and human studies had already suggested meningeal lymphatics might live.
The finding should not be read as “MRI photographed tiny lymphatic vessels.” The more careful claim is that MRI detected macromolecule-rich brightness patterns in expected drainage territories, and researchers interpreted those patterns as lymphatic structures or lymphatic fluid.
Venous Sinuses and Cranial Nerves Formed Two Drainage Maps
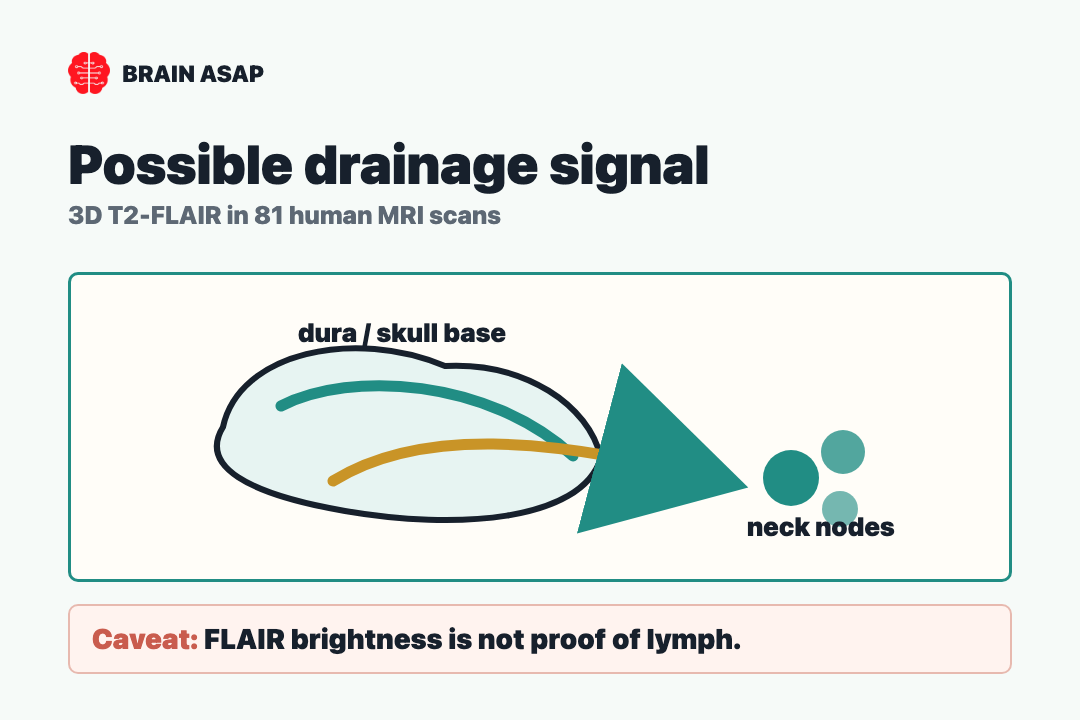
The reported map had two main compartments. The dorsal compartment ran along dural venous structures, including the superior sagittal sinus, straight sinus, confluence, transverse-sigmoid junctions, sigmoid sinuses, jugular veins, and posterior foramen magnum.
The ventral compartment followed skull-base anatomy. Readouts appeared around the anterior cranial fossa, optic groove, Meckel’s cave, internal auditory canal, jugular foramen, and cranial nerve complexes near the route from the skull base into the neck.
The two-route anatomy is informative because animal studies often emphasize ventral skull-base drainage, while earlier human imaging had focused more on parasagittal and dural-sinus regions. Albayram’s team argued that humans may need both routes: one along venous sinuses and another along cranial nerves.
- Dorsal route: Bright readouts clustered near venous sinuses and jugular structures, suggesting a pathway from the dura toward deep cervical lymph nodes.
- Ventral route: Bright readouts appeared around cranial nerve exits and skull-base foramina, suggesting a nerve-associated drainage corridor into the neck.
- Aging measure: Researchers compared readout-to-thickness ratios in younger and older participants.
- Validation need: Follow-up work needs tracer, histology, and artifact-control evidence.
The map becomes more than an atlas if these readouts really mark brain-border drainage.
In that case, they can help translate the glymphatic and meningeal lymphatic literature from mice into living humans.
If they do not, they still expose how hard it is to infer microscopic immune-fluid pathways from clinical MRI brightness.
An 81-Subject MRI Cohort Made Aging the Main Biological Test
The study included 81 people scanned over a 13-month clinical window for epilepsy history or suspected epilepsy, after exclusions for contrast exposure, poor motion quality, surgery, stroke, mass lesions, severe atrophy, status epilepticus, and other factors that could distort normal anatomy.
The mean age was 41.7 years, with a range from 15 to 80.
The cohort let researchers compare younger and older participants.
They used age 50 as a threshold, creating a younger group of 52 people and an older group of 29 people.
The older group had expected brain-aging markers: lower gray and white matter volumes and higher CSF volume.
The lymphatic readout moved too.
Age over 50 was associated with lower dorsal and ventral readout-to-thickness ratios after adjustment for sex and intracranial volume.
The reported odds ratios were 2.9 for the dorsal ratio and 8.3 for the ventral ratio, with the ventral association especially strong.
Researchers interpreted lower readout-to-thickness ratios as possible evidence of reduced drainage efficiency.
By their imaging metric, the channels looked thicker but less efficient, while cervical lymph nodes looked smaller.
The pattern fits a “backup in the drainage system” story, but it remains an imaging interpretation, not a direct flow measurement.
The age pattern was not identical across the two routes.
Age correlated strongly with dorsal thickness, while the ventral readout relationship was more complicated.
Age was positively correlated with mean dorsal thickness and dorsal brightness normalized to white matter, and with mean ventral thickness, but not with ventral brightness normalized to white matter after adjustment.
The FLAIR Criticism Is Not a Footnote
The main caveat is not just “small sample” or “retrospective design.” It is specificity. FLAIR hyperintensity has many possible causes, especially near the skull base and parasagittal dura, where CSF flow artifacts, venous structures, arachnoid granulations, dural channels, and protein-rich fluid can all complicate interpretation.
In a 2023 critique, Ringstad and Eide argued that bright FLAIR readout outside the brain is not enough to identify lymph. Their critique was pointed: some skull-base brightness could be CSF flow artifacts, and parasagittal brightness could reflect non-lymphatic dura or CSF-related structures.
Albayram’s team replied that they were not claiming to directly see lymphatic vessels themselves.
They argued that the brightness may represent waste-rich or macromolecule-rich fluid densities in lymphatic structures, and they pointed to histology and later contrast-based work as partial support for some of the proposed drainage anatomy.
The exchange changes how this evidence should be framed.
The finding is not a settled clinical biomarker.
It is a provocative imaging hypothesis: 3D FLAIR may reveal meaningful drainage-related brightness, but the field still needs prospective validation, ideally with converging MRI sequences, tracer studies, histology, and careful artifact controls.
FLAIR Drainage Readouts Could Become Research Tools for Alzheimer’s and Neuroinflammation
The larger stakes are real.
Brain clearance systems are now tied to questions about sleep, aging, traumatic brain injury, multiple sclerosis, Parkinson’s disease, Alzheimer’s disease, and neuroinflammation.
A human-friendly imaging marker would be valuable because most mechanistic drainage work still comes from animal models or invasive methods.
If meningeal lymphatic output weakens with age, it could help explain why waste clearance, immune surveillance, and inflammatory signaling change across the lifespan.
The paper’s aging pattern points in that direction, but it does not prove a disease mechanism.
An anatomical readout is not a dementia test, and a drainage hypothesis is not a treatment target by itself.
The method is most informative as a research tool.
It can help scientists ask whether drainage-related MRI readouts differ in Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, traumatic brain injury, or normal aging.
It can also force the field to define what counts as evidence: brightness, direction of flow, tracer movement, lymphatic markers, or some combination of all four.
A better version of this research program would pair non-contrast FLAIR with contrast-enhanced slow-flow imaging, standardized posture controls, test-retest reliability, blinded multi-rater anatomy calls, and cohorts that are not selected from epilepsy imaging workflows.
That would make the method less vulnerable to the charge that it is seeing artifacts and naming them anatomy.
The MRI Map Needs Validation Before It Becomes a Diagnosis
The best-supported interpretation is not “MRI has solved brain lymphatics.” It is this: a non-contrast MRI sequence produced a plausible two-route map of human brain-border drainage, linking dural venous regions, cranial nerve exits, vascular neck spaces, and cervical lymph nodes in living people.
The least defensible interpretation would be to turn that map into a diagnostic promise.
Researchers did not show that these readouts diagnose Alzheimer’s disease, predict cognitive decline, measure sleep-dependent glymphatic flow, or respond to treatment.
They also did not settle whether every bright structure they traced is truly lymphatic.
Still, the paper gives researchers a way to test a hidden system in living people.
A drainage pathway that cannot be seen is hard to connect to human disease.
A disputed but measurable MRI readout is at least a starting point, as long as the next studies treat it as a hypothesis to validate rather than a biomarker to sell.
Citation: DOI: 10.1038/s41467-021-27887-0. Albayram et al.. Non-invasive MR imaging of human brain lymphatic networks with connections to cervical lymph nodes. Nature Communications. 2022;13:203.
Study Design: Retrospective human neuroimaging anatomy study using non-contrast 3D T2-FLAIR MRI on a 3-T scanner, plus albumin phantom work to test signal behavior in protein-rich fluid.
Sample Size: 81 human subjects: 45 females and 36 males, mean age 41.7 years, range 15-80. Age analyses compared 52 participants younger than 50 with 29 participants older than 50.
Key Statistic: Age over 50 was associated with lower dorsal and ventral readout-to-thickness ratios after adjustment for sex and intracranial volume; reported odds ratios were 2.9 for dorsal and 8.3 for ventral measures.
Caveat: A 2023 critique challenged the interpretation of hyperintense FLAIR brightness as lymph, arguing that artifacts and non-lymphatic structures can produce similar brightness. The authors replied that the brightness should be understood as putative macromolecule-rich drainage-related brightness, not direct vessel visualization.