TL;DR: Fragile X is usually framed as a neuronal disease. A new mouse study points the finger somewhere else: an overactive BMP signaling program inside astrocytes. Switching it off in glial cells alone — by deleting Smad4 — reduced sound-triggered seizures and partially restored synaptic activity in the auditory cortex.
Key Findings
- The intervention was glial, not neuronal: Smad4 was conditionally deleted in astrocytes only — testing whether astrocytes themselves carry part of the fragile X phenotype.
- Audiogenic seizure severity dropped in male FXS mice: A behaviorally relevant, sensory-triggered seizure phenotype improved after astrocyte-only BMP suppression.
- BMP signaling was elevated in fragile X astrocytes to begin with: The pathway was running hot in the disease state — making it a plausible upstream control point, not just another downstream readout.
- Auditory cortex synaptic activity partly recovered: The molecular fix translated to circuit-level change in the sensory region tied to the behavioral readout.
- Astrocyte transcriptomes and proteomes shifted toward normal: Disrupted metabolic, secretory, secreted-protein, and membrane-protein programs were mitigated when BMP signaling was suppressed.
- Sex-specific effect: The seizure rescue was reported in male FXS mice — female effects need direct testing in follow-up work.
Source: Nature Communications (2026) | Deng et al.
Fragile X syndrome has a tidy origin story. Loss of FMRP — a protein that helps neurons mature, regulate synapses, and stay calibrated — drives the disease. The textbook treatment ambition has been to repair what FMRP loss does to neurons.
This study widens the picture. The most active driver of fragile X dysfunction in this experiment was not in neurons at all. It was in the astrocytes sitting next to them — and changing one signaling pathway inside those support cells was enough to move the disease.
The Astrocyte Hypothesis Is Older Than the Drugs
Astrocytes are the star-shaped support cells that surround synapses, manage local ion and neurotransmitter balance, supply metabolic backup, and release signaling molecules that tell synapses when to form, stabilize, or get pruned. They are not passive scaffolding. They are an active part of how circuits are tuned during development and maintained afterward.
Neurodevelopmental disorders have spent two decades being studied mostly through neurons because that is where the symptoms feel like they should live. The astrocyte story has been catching up — and fragile X is a useful test case because the circuit deficits are well-mapped and the sensory phenotypes are easy to measure.
The pathway this paper went after is bone morphogenetic protein signaling — usually shortened to BMP. BMPs are best known as developmental patterning signals, but they also influence cell identity and gene expression in mature glia. In fragile X astrocytes, BMP signaling was upregulated. That made it a legitimate upstream control point instead of just another downstream abnormality.
Why Smad4 Was the Clean Way to Test the Question
A whole-brain BMP blocker would not have answered the relevant question. BMP signaling runs in many cell types, and a system-wide change would be impossible to attribute. The authors needed a tool that would suppress BMP signaling in astrocytes only — and leave the rest of the brain alone.
That tool was a conditional knockout of Smad4, a central transducer of BMP pathway signals. By deleting Smad4 specifically in astrocytes (a “cKO”), the team turned the question into a clean test: do astrocytes themselves carry part of the fragile X phenotype?
Smad4 is broad enough to cover multiple BMP signals, which is exactly what this kind of question needs. The point was not to identify a perfect drug target. It was to ask whether reducing the pathway inside astrocytes would change the disease state.
The Audiogenic Seizure Test Forced the Pathway to Prove It
Fragile X mice show audiogenic seizures — seizures triggered by sound. The phenotype is not the entire disorder, but it is a behaviorally meaningful readout of sensory excitability. If a molecular rescue is real, it should show up here.
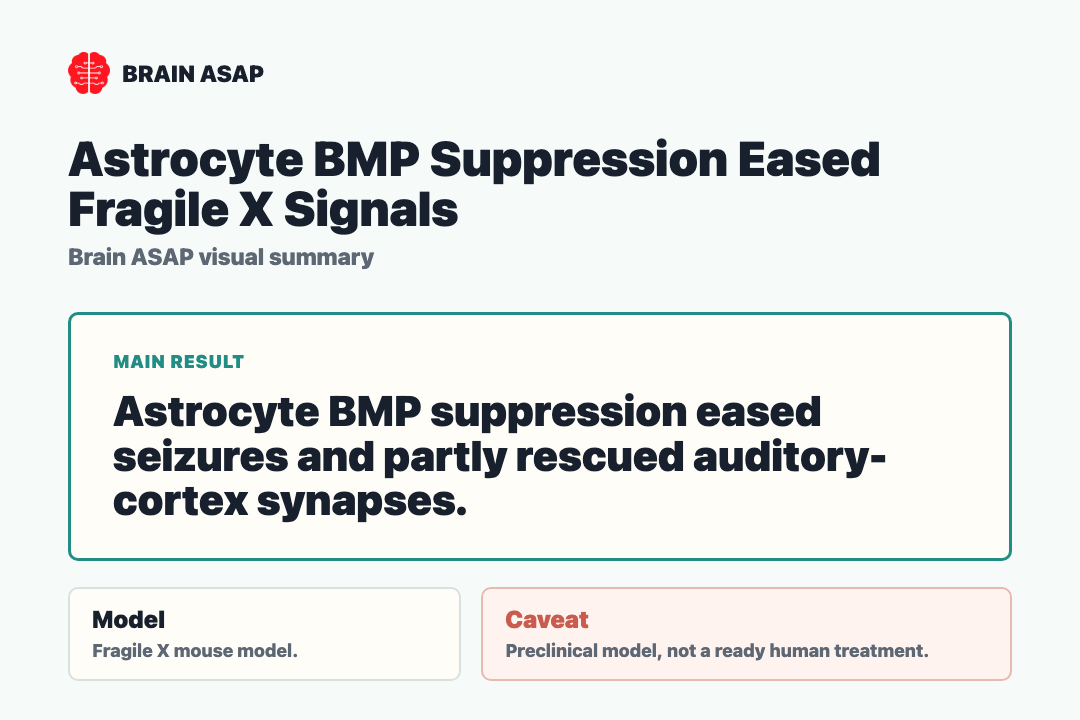
Suppressing astrocyte BMP signaling reduced audiogenic seizure severity in male FXS mice. That is the result that ties an astrocyte molecular pathway to behavior — not just to gene-expression patterns. The intervention was upstream of the synapse, and the effect was downstream at the level of whole-animal behavior.
The sex specificity matters. The seizure improvement was reported in males. Female fragile X biology is known to differ — partial X inactivation, mosaic FMRP expression, distinct sensory profiles — and the paper does not establish that astrocyte BMP suppression behaves the same way in female mice. That needs to be a direct experiment, not an assumption.
Two Layers of Astrocyte Profiling Tell a Layered Story
The team profiled astrocytes with both transcriptomics (RNA — which genes the cell is expressing) and proteomics (protein — what the cell is actually building). The disrupted programs in fragile X astrocytes covered metabolic pathways, secretory machinery, secreted proteins, and membrane proteins. That is the right list for astrocyte biology: these cells regulate energy support, release signals, and shape extracellular environments.
BMP suppression mitigated many of those molecular changes. The RNA and protein layers did not perfectly overlap, which is biologically informative rather than embarrassing. Astrocyte disease can look different depending on whether you measure the messages, the machinery, or the secreted products — and the place where therapeutic leverage actually lives may not be the transcript that first drew attention.
The secretory and membrane-protein findings carry the most weight for the synapse story, because those are the ways astrocytes communicate with the neurons they neighbor. Fix the wrong signal at the cell surface, and the synapse next door cannot help responding.
Partial Rescue at the Synapse Is Still a Real Rescue
The auditory cortex was the circuit that mattered here, both because it processes sound and because it sits in the path of the audiogenic seizure phenotype. After astrocyte BMP signaling was suppressed, synaptic activity in this region partially recovered.
That gives the seizure result a circuit-level companion. The intervention did not only shift astrocyte RNA and protein profiles. It changed neuron-to-neuron communication in the sensory cortex tied to the behavioral readout.
“Partial” is the right word. This is not a full correction of fragile X synapses, and the paper should not be quoted as one. It supports a narrower and more durable claim: astrocyte BMP signaling shapes both glial molecular state and synaptic function in a fragile-X-relevant sensory circuit, and lowering it helps both.
What This Does and Does Not Promise
This is preclinical mouse work using a genetic intervention that no human therapy can directly mimic. Translating it into a clinical tool would mean cell-type-specific delivery, dose control, an appropriate developmental window, and confidence that BMP suppression would not interfere with other repair or developmental functions.
The mechanistic contribution is more concrete than the therapeutic one. Fragile X is often described as a neuronal synapse disorder. This study shows that a fragile X mouse can be measurably moved by changing astrocyte signaling alone — astrocyte molecular profiles, sensory seizure behavior, and synaptic activity all shifted toward normal.
If the pathway holds up in further work, it adds a new layer to the fragile X treatment map. Future therapies will likely need to treat neuron–astrocyte communication as part of the target landscape rather than as supportive scenery. Ignoring astrocytes may mean missing the control point that decides how fragile X circuitry actually behaves.
Two specific follow-ups will determine how far this goes.
- First, which downstream astrocyte function is doing the actual work — metabolic support, secreted signals, synaptic-environment molecules, or all of those at once?
- Second, when does the intervention need to happen? A pathway that helps during early development could be risky to suppress broadly, while a later, narrower window may be safer if it mostly tunes mature astrocyte function. That timing answer will decide whether the right target is Smad4 itself, a specific downstream astrocyte program, or a single secreted molecule released into the synaptic environment.
Citation: Deng et al. Suppression of astrocyte BMP signaling improves molecular signatures and functional deficits in a fragile X syndrome mouse model. Nature Communications. 2026. DOI: 10.1038/s41467-026-71919-6
Study Design: Fragile X mouse mechanism study with astrocyte-specific Smad4 conditional knockout, transcriptomics, proteomics, audiogenic seizure testing, and auditory-cortex synaptic-activity analysis.
Sample: Fragile X mouse model with astrocyte-specific genetic manipulation.
Key Result: Astrocyte Smad4 cKO reduced audiogenic seizure severity in male FXS mice and partially rescued auditory-cortex synaptic activity.
Boundary: Mouse mechanism study; human translation requires safe timing, cell specificity, and dose control.