TL;DR: Microplastics circulating in your blood may accelerate Alzheimer’s and Parkinson’s disease through six shared pathways—including blood-brain barrier breakdown, chronic inflammation, and oxidative stress.
You probably know plastic pollution is everywhere. Less obvious: tiny plastic fragments are crossing into your brain, accumulating in brain tissue, and potentially triggering neurodegenerative disease.
A new review in Molecular and Cellular Biochemistry reveals how microplastics share the same disease-driving mechanisms as Alzheimer’s and Parkinson’s, raising urgent questions about long-term neurological risk.
Key Findings
- 121,000 microplastics ingested annually: Adults may ingest up to 121,000 microplastics per year from contaminated food, water, and air—particles that resist breakdown and persist in the body.
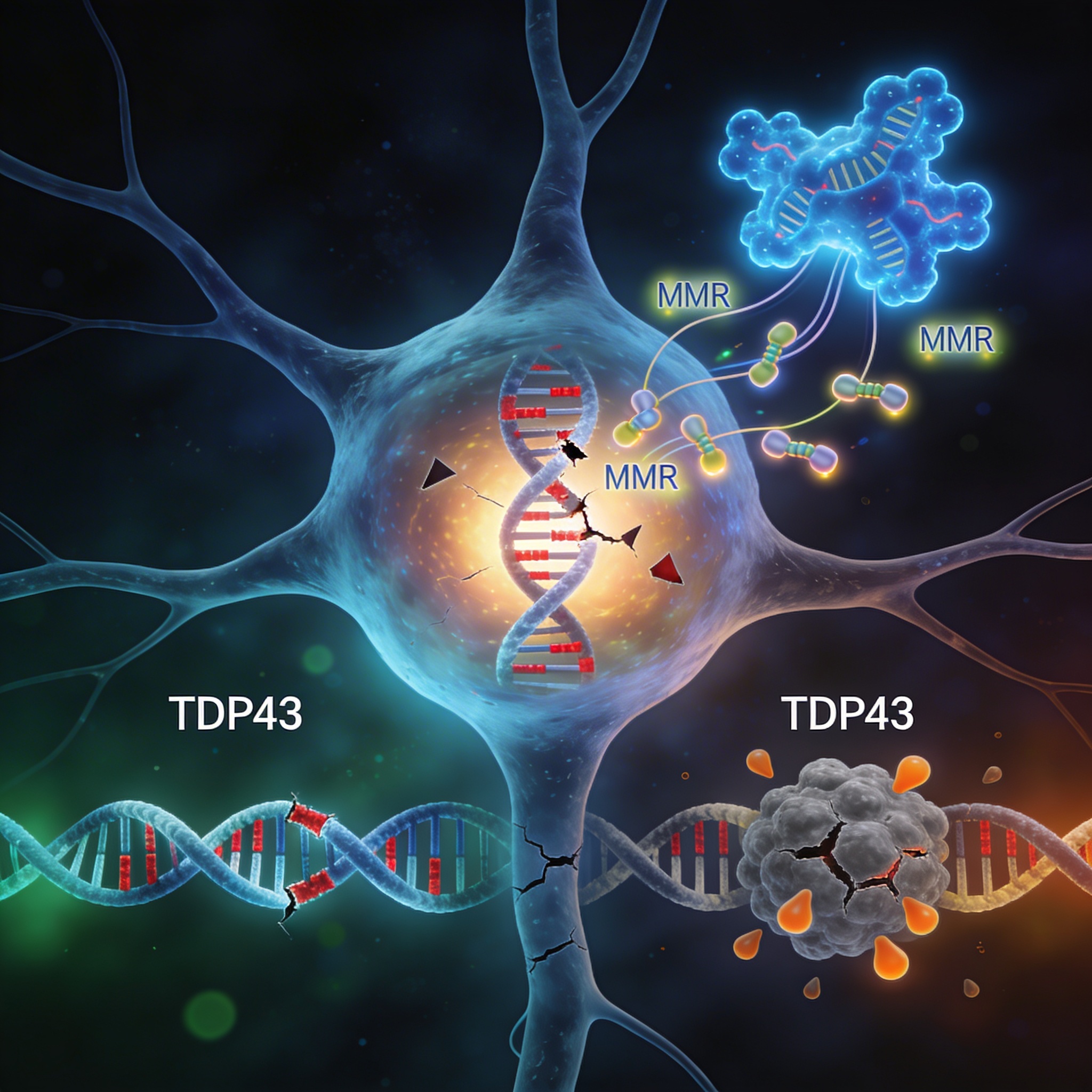
- Blood-brain barrier permeability increased: Microplastics cross the BBB and downregulate tight junction proteins (claudins, occludin, ZO-1), enabling harmful substances to infiltrate the central nervous system.
- Neuroinflammation via microglial activation: Microplastics trigger microglia to release pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) and activate critical immune signaling pathways (TLR4, ERK, NFκB, MYD88).
- Oxidative stress and ROS production: Microplastics generate reactive oxygen species (ROS), depleting antioxidant defenses (SOD, CAT, GST) and impairing mitochondrial function—a hallmark of neurodegeneration.
- Mitochondrial dysfunction and energy collapse: Microplastics disrupt ATP synthesis and oxidative phosphorylation, reducing neuronal energy and triggering apoptosis and tau aggregation.
- Six shared mechanisms with Alzheimer’s and Parkinson’s: BBB disruption, chronic neuroinflammation, oxidative stress, mitochondrial dysfunction, impaired autophagy, and epigenetic modifications all linked to microplastic exposure.
Source: Molecular and Cellular Biochemistry (2026) | Chi Wang Siu et al.
How Microplastics Enter Your Body—and Reach Your Brain
Microplastics come from three routes: ingestion (food and water), inhalation (air), and dermal contact (creams, fabrics). Seafood carries particularly high loads—polyester fibers from textiles and paint particles from corroded gear end up in marine systems and eventually on your plate.
Once in the bloodstream, smaller particles (less than 0.5 µm) cross biological barriers through phagocytosis, transendothelial transport, and lipid-bilayer diffusion. Brain tissue accumulation occurs via receptor-mediated uptake and cerebrospinal fluid distribution. A 2024 human study found the highest microplastic concentrations in the frontal cortex—a region critical for cognition and executive function.
The Blood-Brain Barrier: The First Line of Defense Breaks Down
The blood-brain barrier (BBB) is a selective gate, preventing most toxins from entering the central nervous system. Microplastics directly compromise this boundary. A 2025 study by Kim et al. demonstrated that microplastic exposure downregulates tight junction proteins—claudins, occludin, and zonula occludens-1 (ZO-1)—reducing BBB integrity and increasing permeability by up to 4.5-fold.
This breach activates a cascade: compromised BBB allows pro-inflammatory cytokines (TNF-α, IL-6) and activated microglia to infiltrate brain tissue. The result is sustained neuroinflammation—exactly what precedes Alzheimer’s and Parkinson’s pathology. Early BBB breakdown is now recognized as a critical feature in disease onset and progression.
Chronic Neuroinflammation: Microglial Activation and Cytokine Storm
Microplastics are recognized as foreign invaders by resident immune cells. Microglia engulf the particles through phagocytosis, which triggers morphological polarization and upregulation of pro-inflammatory markers (M1 phenotype). A 2022 study showed microplastic-exposed microglia released elevated levels of TNF-α, IL-1β, and IL-6—cytokines linked to neuronal death and cognitive decline.
The inflammatory cascade extends to critical immune signaling pathways. Microplastic exposure activates TLR4, ERK, NFκB, and MYD88 proteins, amplifying the production of destructive chemokines (CXCL10, MCP-1) that recruit more immune cells and sustain neuroinflammation. Astrocytes (support cells) also become activated, contributing to a self-perpetuating cycle of inflammation that damages neural integrity.
Oxidative Stress: The Free Radical Problem
Microplastics generate reactive oxygen species (ROS) through multiple mechanisms. Internalized particles disrupt mitochondrial membranes, increasing ROS production and overwhelming the cell’s antioxidant defenses (superoxide dismutase, catalase, glutathione S-transferase). A 2024 mouse study found that polyethylene microplastic exposure reduced SOD and CAT activity while elevating malondialdehyde (MDA)—a marker of lipid peroxidation and oxidative damage.
The damage cascades: elevated oxidative stress promotes amyloid-beta aggregation and tau hyperphosphorylation—the hallmark protein tangles of Alzheimer’s disease. Oxidative stress also impairs proteasomal and lysosomal function, preventing neurons from clearing misfolded proteins and leading to their accumulation.
Mitochondrial Dysfunction: Energy Failure in Brain Cells
Neurons are energy hogs, and mitochondria are their power plants. Microplastics localize to mitochondria and disrupt oxidative phosphorylation—the process that generates ATP, the cell’s energy currency. Once inside, microplastics depolarize mitochondrial membranes, reducing the proton gradient needed for ATP synthesis and promoting excess ROS production.
The consequences are severe: impaired ATP production starves neurons of energy for basic functions. This energy collapse activates apoptosis (programmed cell death) and fails to power the protein-clearance pathways that normally prevent tau and amyloid accumulation. Mitochondrial dysfunction and energy failure are central features of both Alzheimer’s and Parkinson’s pathology.
Autophagy Breakdown and Protein Accumulation
Cells rely on autophagy—a housekeeping system that degrades misfolded proteins and damaged organelles—to maintain health. Microplastics interfere with both lysosomal and autophagy pathways. A 2022 study demonstrated that polystyrene microplastics inhibited autophagy-dependent ferritophagy and apoptosis in neuronal cells, preventing the clearance of iron-laden ferritin and misfolded proteins.
This impairment is especially dangerous in neurons. When autophagy fails, alpha-synuclein (the protein that misfolds in Parkinson’s disease) accumulates into toxic Lewy bodies. In Alzheimer’s disease, failed autophagy allows amyloid-beta plaques and tau tangles to build up unchecked. The evidence suggests that microplastic-induced autophagy dysfunction may be a critical link between environmental exposure and neurodegeneration.

Epigenetic Chaos: DNA Damage and Gene Dysregulation
Beyond direct toxicity, microplastics alter the chemical switches that control gene expression. A 2022 study found that zebra fish embryos exposed to microplastics showed altered DNA methylation and reduced expression of maintenance de novo DNA methyltransferases—enzymes that preserve proper gene silencing.
Exposure dysregulates critical antioxidant and neuroprotective genes: reduced expression of SOD1, CAT, GPX1a, and HSP70 leaves neurons vulnerable to oxidative attack. These same epigenetic changes have been documented in Alzheimer’s disease patients, suggesting that microplastic-induced epigenetic alterations may accelerate disease progression.
Two Specific Diseases, One Environmental Culprit
The review identified six mechanisms specific to Alzheimer’s disease: BBB disruption, chronic neuroinflammation, oxidative stress, mitochondrial dysfunction, impaired autophagy, and epigenetic modifications. Each pathway is independently linked to amyloid-beta accumulation and tau pathology.
For Parkinson’s disease, six additional mechanisms emerged: BBB disruption, oxidative stress in dopaminergic neurons (particularly vulnerable due to dopamine metabolism), mitochondrial dysfunction, neuroinflammation, alpha-synuclein aggregation, and gut-brain axis disruption. The emphasis on dopaminergic vulnerability is critical: dopamine metabolism generates high ROS levels, making these neurons especially susceptible to microplastic-induced oxidative damage.
Alzheimer’s Disease: The Amyloid-Tau Trap
Alzheimer’s disease is marked by amyloid-beta plaques and tau tangles in the brain. Microplastic exposure promotes this cascade through multiple routes. Oxidative stress accelerates amyloid processing and tau hyperphosphorylation. Chronic neuroinflammation activates the NLRP3 inflammasome, releasing IL-1β and IL-18—both linked to amyloid accumulation.
Impaired mitochondrial function fails to generate sufficient ATP for autophagy, allowing toxic protein aggregates to accumulate unchecked. BBB breakdown permits peripheral neurotoxic substances and microglial activation to amplify brain pathology. Epigenetic changes silence protective genes while upregulating those involved in inflammation and oxidative stress. Together, these mechanisms form a perfect storm for AD pathogenesis.
Parkinson’s Disease: A Dopaminergic Collapse
Parkinson’s disease strikes dopamine-producing neurons in the substantia nigra. This brain region is uniquely vulnerable to oxidative stress because dopamine metabolism itself generates reactive oxygen species. Microplastic exposure amplifies this vulnerability: elevated ROS production combines with reduced antioxidant defenses to overwhelm dopaminergic neurons.
Microplastics also promote alpha-synuclein aggregation—the misfolded protein that forms Lewy bodies, the pathological hallmark of Parkinson’s. A 2023 study found that exposure to amniotic nanoparticles accelerated alpha-synuclein fibril formation and neurotoxicity. Additionally, microplastics disrupt the gut microbiota, altering the production of protective metabolites and promoting dysbiosis-driven inflammation that propagates to the brain through the vagus nerve.
The Gut-Brain Connection in Parkinson’s
Emerging evidence links gut dysbiosis to Parkinson’s progression. Microplastics damage intestinal barrier integrity, allowing bacterial lipopolysaccharides and other pro-inflammatory compounds to enter the bloodstream. These activate systemic and neuroinflammation, promoting alpha-synuclein aggregation.
The vagus nerve—the major communication cable between gut and brain—appears to be a conduit for pathological alpha-synuclein, potentially initiating or accelerating disease. Clinical observations show that Parkinson’s patients often have dysbiosis and altered microbial-derived metabolite profiles, aligning with the brain’s burden of microplastic-associated damage.
Critical Limitations and Unanswered Questions
This review synthesizes preclinical and animal evidence, but human clinical data remain sparse. Most studies use polystyrene microplastics (PS-MPs) at high experimental concentrations, which may not perfectly mirror real-world exposure patterns. Long-term human cohort studies tracking microplastic ingestion and cognitive decline are urgently needed.
Regional brain accumulation patterns are still poorly understood. The frontal cortex shows highest concentrations, but whether other brain regions (hippocampus, substantia nigra) accumulate microplastics differently remains unclear. Additionally, most research is short-term, measuring responses over weeks or months—not the years or decades required for neurodegeneration to manifest clinically.
What This Means for Public Health
If microplastics genuinely contribute to neurodegeneration, the implications are staggering. Over 57 million people globally live with dementia, with Alzheimer’s disease accounting for 60-70% of cases. Parkinson’s affects over 10 million people worldwide. Even a modest increase in risk from environmental plastic exposure would represent a massive public health burden.
The evidence points to the urgent need for longitudinal human studies, standardized measurement protocols, and stricter regulation of plastic production and waste management. Reducing personal exposure—through dietary choices, filtering water, minimizing inhalation of synthetic textile fibers—may offer protection while we await definitive causal evidence.
Citation: Siu ACW, Paudel KR, Singh G, et al. Do microplastics play a role in the pathogenesis of neurodegenerative diseases? Shared pathophysiological pathways for Alzheimer’s and Parkinson’s disease. Molecular and Cellular Biochemistry. 2026;481:669-694. DOI: 10.1007/s11010-025-05428-3
Authors’ affiliations: Graduate School of Health, University of Technology Sydney; Centre for Inflammation, School of Life Sciences, Faculty of Science, University of Technology Sydney; Woodcock Institute of Medical Research, Macquarie University; Uttaranchal University, Dehradun; Centre for Research Impact & Outcome, Chitkara University; Centre of Medical and Bio-allied Health Sciences Research, Ajman University; School of Pharmaceutical Sciences, Lovely Professional University; Faculty of Health, Australian Research Centre in Complementary and Integrative Medicine, University of Technology Sydney; Department of Drug Discovery & Development, Auburn University; School of Life Sciences, University of Technology Sydney