TL;DR: Obesity and depression form a pathophysiological trap: inflammatory cytokines and dysbiotic bacteria from obesity trigger neuroinflammation and mood disruption, while depression’s behavioral changes and HPA axis dysfunction worsen obesity.
Obesity and depression are twin epidemics in the modern world, and they’re not independent. People with obesity are 1.5 to 2 times more likely to develop depression than lean peers. The reverse is equally true: depressed patients gain weight more readily. For decades, the assumption was simple—weight gain breeds shame and inactivity, or depression’s lethargy leads to overeating.
But the truth is stranger. Obesity and depression are locked in a bidirectional relationship where each feeds the other through chronic inflammation, gut dysbiosis, hormonal imbalance, and central nervous system dysfunction.
Breaking this cycle requires treating both conditions simultaneously. Weight loss alone or antidepressants alone often fail because they ignore the shared biological pathways that keep patients trapped.
Key Findings
- Shared inflammatory pathways: Obesity and depression share common inflammatory pathways: elevated pro-inflammatory cytokines (IL-6, TNF-α, IL-1β) are found in both conditions and bidirectionally promote symptom worsening.
- Gut microbiota dysfunction: The gut microbiota acts as a critical intermediary; obesity-associated dysbiosis alters short-chain fatty acid (SCFA) production and tryptophan metabolism, disrupting the microbiota-gut-brain (MGB) axis and impairing mood regulation.
- Intestinal permeability: Chronic low-grade inflammation in obesity increases intestinal permeability, allowing lipopolysaccharides (LPS) and bacterial antigens to cross the blood-brain barrier and activate neuroinflammatory cascades in the limbic system.
- HPA axis dysregulation: Hypothalamic-pituitary-adrenal (HPA) axis dysregulation is common to both: obesity blunts cortisol responses, while depression heightens them, creating reciprocal metabolic stress.
- Leptin resistance: Leptin resistance in obesity impairs leptin’s neuroprotective and anti-inflammatory signaling in the brain, exacerbating depression risk.
- Early life adversity: Early life adversity and stress amplify both obesity and depression risk, suggesting shared developmental vulnerability.
Source: International Journal of Molecular Sciences (2025) | Monsalve et al.
One Condition Promotes the Other: Bidirectional Risk
The pattern is well-documented and bidirectional. Depressed patients also gain weight more readily and struggle to lose it. Standard explanations—shame, inactivity, lethargy—capture only the surface. The deeper mechanism is biochemical and neuroinflammatory.
The Inflammatory Echo Chamber
Obesity is a chronic inflammatory state. The core mechanism involves:
- Adipose tissue secretion: Visceral fat releases pro-inflammatory cytokines (TNF-α, IL-6, IL-1β)—the same cytokines elevated in depressed patients
- Blood-brain barrier penetration: These cytokines cross into the brain and trigger neuroinflammatory cascades that impair the hippocampus and prefrontal cortex
- Depressive amplification: Depressed mood intensifies stress signaling and amplifies inflammatory output
- Bidirectional feedback: The two conditions feed each other through inflammation like an echo chamber
The Microbiota Trap
Obesity disrupts the gut microbiota through a cascade of failures. Dysbiotic communities produce fewer short-chain fatty acids (SCFAs), which normally nourish the intestinal barrier and regulate immune tolerance.
Without adequate SCFAs, tight junctions weaken and intestinal integrity fails. Lipopolysaccharides—toxic bacterial wall components—leak into the bloodstream, triggering immune activation and neuroinflammation.
Depression compounds the problem by altering eating patterns and stress signaling. Each condition worsens the other’s microbiota in a vicious spiral.
The Gut-Brain Axis: Where Obesity Breaks Mood Regulation
The microbiota-gut-brain axis is how the intestinal ecosystem talks to the brain. Healthy microbes produce neurotransmitters and tryptophan metabolites that influence mood. Dysbiotic communities fail at this. Obesity-associated dysbiosis shows a specific pattern of microbial loss and metabolic dysfunction:
- Species reduction: Reduced butyrate-producing species like Faecalibacterium prausnitzii
- SCFA cascade: Fewer SCFAs mean weaker immune tolerance and heightened reactivity to gut antigens
- Tryptophan rerouting: Dysbiotic communities shunt tryptophan toward inflammatory kynurenine derivatives instead of mood-protective compounds
- Neuropsychiatric impact: Elevated kynurenine-to-tryptophan ratios are associated with depression and anxiety
A depressed patient with obesity faces a double hit: low SCFA production and dysregulated tryptophan metabolism, both hammering mood from within the gut.
The intestinal barrier itself becomes compromised. Tight junction proteins are weakened by low butyrate and sustained LPS exposure.
Dysbiosis plus chronic inflammation allow bacterial components to cross into circulation and trigger neuroinflammation in the brain.
Leptin Resistance: The Brain’s Appetite Regulator Stops Listening

Leptin, the “satiety hormone,” is more than appetite control. It’s a cytokine with neuroprotective properties. In healthy individuals, leptin suppresses inflammatory signaling, enhances synaptic plasticity, and supports mood regulation.
In obesity, leptin levels soar yet the brain becomes insensitive—leptin resistance develops. The brain loses its neuroprotective effects: neuroinflammation intensifies, depression risk rises, and appetite circuits become hyperactive, perpetuating weight gain.
Depression then exacerbates leptin resistance by amplifying inflammatory cytokine production, which further impairs leptin receptor function. The brain’s appetite and mood regulator has effectively gone deaf.
HPA Axis Dysfunction: Stress Hormones Gone Awry
The HPA axis controls stress responses and metabolism. In obesity and depression, it becomes severely dysregulated in opposing directions:
- Obesity pattern: Despite chronic inflammatory stress, cortisol responses are often blunted, failing to suppress ongoing inflammation
- Depression pattern: Hyperactive HPA axis with excessive cortisol secretion
- Combined effect: When a patient has both conditions, obesity’s blunted response impairs anti-inflammatory signaling while depression’s hyperactive HPA adds metabolic stress
Chronic cortisol dysregulation promotes belly fat accumulation and impairs the mood-regulating hippocampus—worsening depression. The cycle tightens.
Inflammation Across the Lifespan: Early Roots
Vulnerability to the obesity-depression link begins early. Adverse childhood experiences and early-life stress prime the developing brain and body through multiple pathways.
Early stress primes the immune system toward elevated baseline inflammation and alters microbiota composition. It also reduces children’s capacity to handle future stressors.
The microbiota-gut-brain axis is particularly vulnerable: early dysbiosis during sensitive developmental periods can permanently alter mood-regulating circuits, setting the stage for both obesity and depression later in life.
Interventions targeting the microbiota-brain axis—dietary changes, stress reduction—may be most effective when started early, before the obesity-depression cycle becomes entrenched.
Breaking the Cycle: Treatment Implications
Treating obesity alone often fails because depressive symptoms resurface and sabotage adherence.
Antidepressants alone may not lift mood if obesity-driven inflammation continues.
The evidence points toward integrated treatment that addresses all pathways simultaneously:
- Lifestyle interventions: Exercise, dietary changes (anti-inflammatory patterns), and sleep optimization that simultaneously reduce inflammation and improve metabolic health
- Psychotherapy: Addressing depression and stress-driven eating patterns
- Microbiota restoration: Dietary fiber, fermented foods, and targeted probiotics to restore dysbiotic communities
- Simultaneous interruption: Dampen inflammation, restore the microbiota, normalize stress hormone signaling, and rebuild brain resilience at once
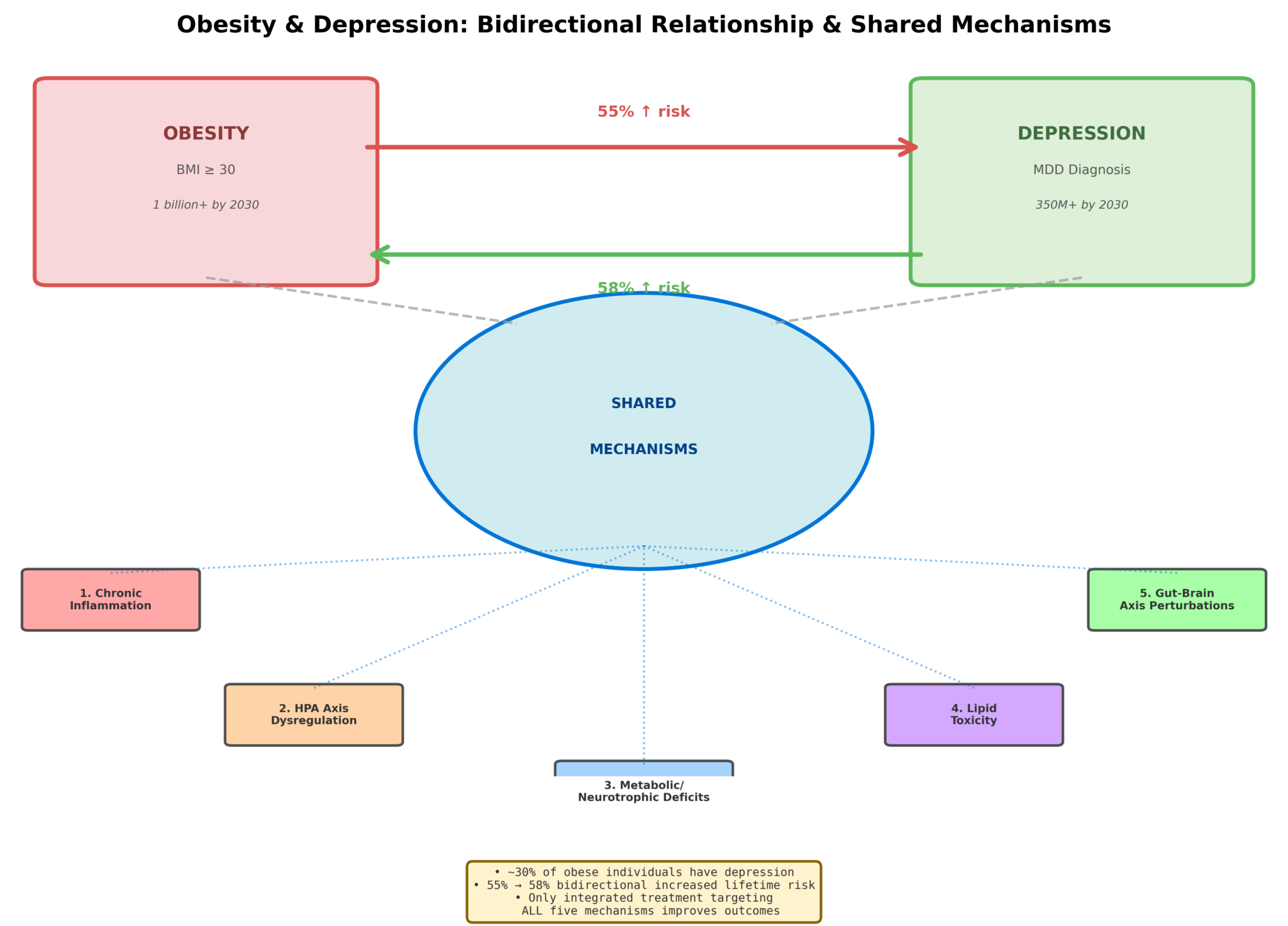
Pathophysiological intersections between obesity and depression: shared inflammatory, metabolic, and neuroendocrine mechanisms create bidirectional risk amplification.
Citation: Monsalve, G., et al. (2025). The pathophysiotoxic relationship between obesity and depression: Inflammatory, metabolic, and neuroendocrine mechanisms. International Journal of Molecular Sciences, 26, 11590. DOI: 10.3390/ijms26211590
Key mechanisms reviewed: Pro-inflammatory cytokines (TNF-α, IL-6, IL-1β), microbiota-gut-brain axis dysfunction, leptin resistance, HPA axis dysregulation, intestinal barrier permeability, and early-life adversity as shared risk factor.