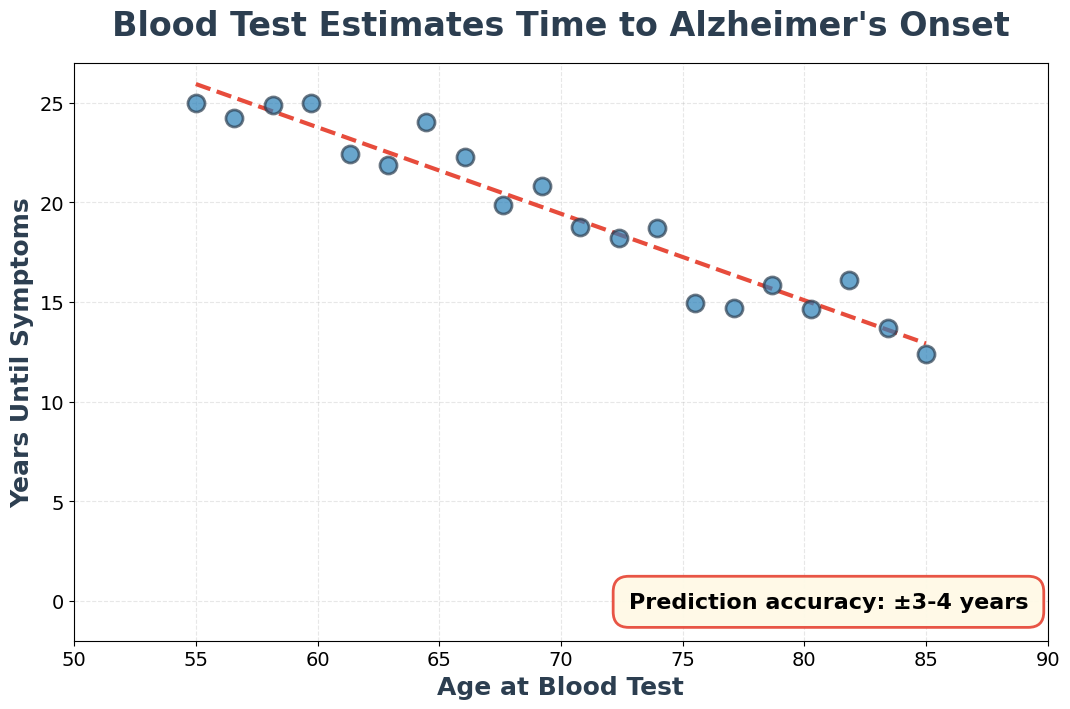
TL;DR: A blood test measuring phosphorylated tau-217 can predict when cognitively normal people will develop Alzheimer’s symptoms with a 3.0-3.7 year margin of error, potentially transforming how researchers identify candidates for preventive clinical trials.
For decades, Alzheimer’s disease remained invisible until symptoms emerged. Brain scans could show amyloid plaques and tau tangles, but only in people already experiencing cognitive decline. The tragedy: by the time symptoms appear, the disease has ravaged the brain for 10-20 years. Researchers needed a way to identify who would develop symptoms before they arrive. Now, a blood test may have solved that problem.
Key Findings
- 3.0-3.7 year prediction window: The p-tau217 blood test estimates when cognitively unimpaired individuals will develop symptomatic Alzheimer’s disease with a median absolute error of just 3-4 years in both the Knight ADRC and ADNI cohorts.
- Age dramatically affects timing: A 60-year-old with elevated p-tau217 faces a 20.5-year median wait until symptoms; a 90-year-old faces only 5.1 years, revealing how age shapes disease trajectory.
- Single blood marker efficiency: Unlike expensive PET imaging, plasma p-tau217 provides temporal staging of Alzheimer’s pathology using a single, accessible blood test from standard clinical assays.
- Robust cross-cohort validation: Clock models trained on the Knight ADRC cohort (258 individuals) replicated strongly in the independent ADNI cohort (345 individuals), with adjusted R² values around 0.73-0.81.
- Alignment with brain pathology: Plasma p-tau217 increases alongside amyloid and tau accumulation across all four biological stages of Alzheimer’s disease, confirming it captures genuine disease progression.
- Clinical trial game-changer: The model identifies symptom onset risk within defined windows, potentially enabling researchers to select participants for preventive trials before cognitive loss begins.
Source: Nature Medicine (2026) | Petersen et al.
The Rise of Blood-Based Biomarkers
Predicting disease onset was always the holy grail of dementia research. Brain imaging—PET scans measuring amyloid and tau—could show pathology, but the technology was expensive, hard to access, and required specialist interpretation.
Enter plasma biomarkers: molecules in your blood that reflect brain pathology. Over the past five years, researchers discovered that phosphorylated tau (p-tau) variants, along with amyloid-beta ratios, correlate with Alzheimer’s pathology. Among these candidates, p-tau217 emerged as a star performer. It’s not just a marker of disease presence; the level changes predictably as the brain’s pathology evolves.
From Detection to Prediction: The Clock Models
The breakthrough came from treating p-tau217 levels as a “clock”—a biological timeline. Researchers took longitudinal data from two large, well-characterized cohorts: the Knight ADRC (258 cognitively unimpaired people with elevated AD biomarkers) and the ADNI (345 people followed over years). They measured p-tau217 values repeatedly over time and asked a simple question: can we estimate how long until someone transitions from cognitively normal to symptomatic AD based on their p-tau217 level?
The answer was yes. Using two independent mathematical approaches (TIRA and SILA), researchers built clock models that converted plasma p-tau217 concentration into an estimated number of years to symptom onset.
The results were striking. Across both cohorts and both modeling methods, the median absolute error—how far off the predictions were from actual symptom onset—hovered at 3.0-3.7 years. That’s tighter than many clinical metrics.
Age Is the Hidden Variable
But here’s where it gets clinically interesting: age changes everything. The researchers stratified individuals by p-tau217 positivity age and looked at progression rates.
A person who becomes p-tau217 positive at age 60 has an estimated 20.5-year median wait until symptoms. That same person at age 70 faces 16.5 years. At 80, it drops to 11.4 years. And at age 90, only 5.1 years. The older you are when pathology begins, the faster symptoms emerge.
This wasn’t unexpected—age-related biological changes accelerate disease progression. But the precision of the prediction is new. Clinicians now have a way to estimate, for a specific patient, how much time they have. For a 75-year-old with newly detected p-tau217 elevation, doctors can tell them, with reasonable accuracy, roughly when cognitive problems may begin.
Alignment With Brain Pathology Stages
One concern with any new biomarker: does it actually track what’s happening in the brain? Researchers addressed this by comparing plasma p-tau217 trajectories to the four biological stages of Alzheimer’s disease defined by amyloid PET, tau PET, and neurodegeneration markers.
Stage A (normal biomarkers) showed low plasma p-tau217 levels. Stage B (amyloid accumulation begins) showed rising p-tau217. Stages C and D (tau spread and neurodegeneration) showed continued increases, tracking the expected pathological progression. The plasma biomarker was doing its job—faithfully reflecting what’s happening in the brain.
Importantly, plasma p-tau217 increased even before tau PET showed significant signal in many individuals. The blood test may be capturing early tau pathology that brain imaging misses, making it even more sensitive for early detection.
From Science to Clinical Trials
Why does this matter? Because preventive trials are the frontier of Alzheimer’s research. Instead of waiting for symptoms to appear and trying to stop progression, researchers now want to treat people who show pathology but remain cognitively normal.
The challenge: how do you enroll the right people? You need cognitively normal individuals with confirmed AD pathology who will likely develop symptoms within the trial timeline. Enroll people too early, and they won’t show enough cognitive decline to measure treatment benefit. Enroll them too late, and the window has closed.
The p-tau217 clock models solve this targeting problem. Researchers can use the plasma test to estimate symptom onset, then preferentially enroll people whose predicted onset falls within the trial period. This dramatically increases the chance of detecting treatment efficacy and reduces the time and cost of large trials.
Limitations and Practical Considerations
The study itself acknowledged important constraints. The p-tau217 clock models only work well within a specific biomarker range (roughly 1.06% to 10.45%). Values outside this band yield unstable predictions. This means the test won’t identify everyone at future risk—only those already on a clear pathological trajectory.
The research also included mostly non-Hispanic white participants, limiting generalizability to other ethnic groups, who may have different disease rates and biomarker dynamics. Participants were largely cognitively normal at enrollment, so the findings don’t necessarily apply to people with existing cognitive impairment.
Additionally, the clock models estimate group-level trends, not individual futures. A predicted 3-4 year window is a median, not a certainty. Some people will develop symptoms sooner; others will remain stable longer.
The Broader Landscape
This paper is part of a larger movement toward blood-based biomarkers in neurodegenerative disease. Other phosphorylated tau variants (p-tau181, p-tau199) and the p-tau217/Aβ42 ratio are also under study. What makes this work distinctive is its focus on temporal prediction—not just detecting pathology, but forecasting when symptoms will arrive.
The approach could eventually extend beyond Alzheimer’s. The same methodology—clock modeling from longitudinal biomarker trajectories—might work for other neurodegenerative diseases with predictable pathological progressions.
What This Means Now
For researchers: plasma p-tau217 clocks offer a practical tool for enriching prevention trials with people most likely to benefit from early intervention. For patients with elevated p-tau217: this test provides something rare—a rough timeline for when cognitive changes might emerge, allowing informed personal planning.
For the field: this work reinforces a fundamental shift. The future of dementia care isn’t about treating symptoms. It’s about detecting pathology early and intervening before the damage is done. Blood tests that predict symptom onset are moving that future closer to reality.
Citation: Petersen RC et al. Predicting onset of symptomatic Alzheimer’s disease with plasma p-tau217 clocks. Nature Medicine. 2026;32:1085-1094. DOI: 10.1038/s41591-026-04206-y
Authors’ affiliations: Department of Neurology, Washington University in St. Louis (Petersen, Fagan); Northern California Institute for Research and Education (Mila-Aloma); Department of Radiology and Biomedical Imaging, University of California, San Francisco (Du-Cunha, Coomararswamy, Du); Wisconsin Alzheimer’s Institute and University of Wisconsin School of Medicine and Public Health (Madison); Knight Alzheimer’s Disease Research Center, Washington University (Morris, McCade); Janssen Pharmaceuticals (Xiong); Center for Biomarker Research in AD, University of Gothenburg (Zetterberg); and additional collaborating institutions across the FNIH Biomarkers Consortium.