TL;DR: In Swedish registry data covering nearly 100,000 people with depression or anxiety, psychiatric hospital visits and sickness absence were 42% lower during semaglutide treatment periods — with comparable reductions in depression, anxiety, and substance-use outcomes.
Key Findings
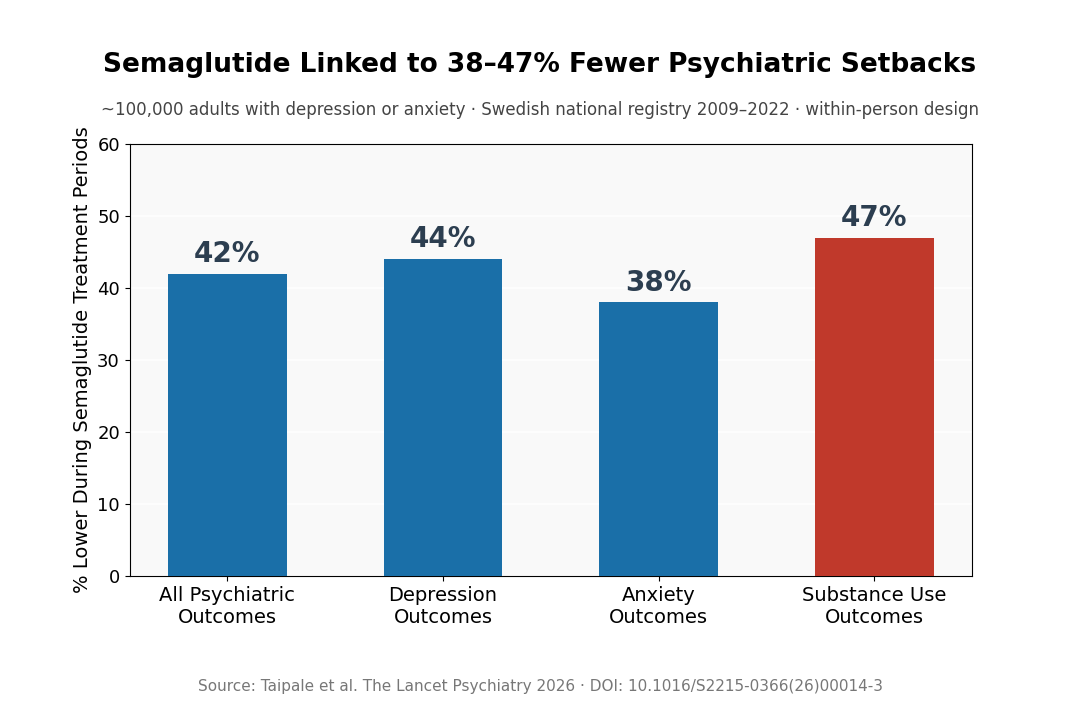
- 42% lower psychiatric care during semaglutide periods: Psychiatric hospital visits and sickness absence episodes dropped 42% when the same individuals were on semaglutide compared to when they weren’t.
- Depression outcomes fell 44%: Depression-related hospital care and sickness absence were 44% lower during semaglutide treatment windows.
- Anxiety outcomes fell 38%: Anxiety-disorder-related severe events followed a similar pattern.
- Substance-use outcomes fell 47%: Hospitalizations and sickness absence tied to substance use were nearly halved during GLP-1 treatment periods.
- Within-person design reduced between-person confounding: The analysis compared each person’s on-drug and off-drug periods rather than GLP-1 users against non-users — a stronger design than a simple cohort comparison.
- Scale: nearly 100,000 people, 13 years of registry data: 2009–2022, Swedish national health registers, including more than 20,000 GLP-1 users with depression or anxiety diagnoses.
Source: The Lancet Psychiatry (2026) | Taipale et al.
GLP-1 receptor agonists — semaglutide (Ozempic, Wegovy), liraglutide — were developed for diabetes and weight loss. But as prescriptions have expanded into people with psychiatric disorders, a different question has become urgent: do these drugs make mental illness better or worse?
The fear was worsening. People with serious depression or anxiety are already metabolically vulnerable, and the nausea, appetite suppression, and body-image changes that come with GLP-1 drugs could plausibly destabilize them. This large Swedish registry study asked that question directly — and found the signal moved the opposite way.
Why a Within-Person Design Matters Here
The study used a within-person comparison: it tracked the same individuals during periods when they were on a GLP-1 drug versus periods when they weren’t. That’s a meaningfully different analysis than comparing GLP-1 users to non-users.
People with psychiatric disorders who end up on semaglutide aren’t a random group. They may have better baseline metabolic control, more clinical contact, higher motivation for treatment, or stronger support systems. A between-person comparison would absorb all those differences and potentially inflate the apparent benefit.
By comparing each person to themselves, the analysis removes stable individual confounders. What remains is whether the same person’s rate of severe psychiatric outcomes differed when they were actively taking the drug. It still leaves room for time-varying confounding — health changes, medication switches, life events — but it’s a stronger foundation than most observational registry studies offer.
The Substance-Use Signal Is the Unexpected Headline
The 42–44% reductions in depression and anxiety events are striking. But the substance-use finding — a 47% drop in hospitalizations and sickness absence tied to substance-use disorders during semaglutide treatment — may be the most scientifically interesting result in the paper.
GLP-1 biology already has traction in addiction research. GLP-1 receptors sit in reward circuitry — the striatum, prefrontal cortex, areas implicated in craving, motivation, and impulsive behavior. Animal studies have shown GLP-1 agonists can reduce alcohol seeking, nicotine preference, and opioid craving. Some human case reports have described unexpected reductions in alcohol intake among GLP-1 users treated for metabolic reasons.
A nearly 50% reduction in substance-use-related severe events in a registry of 100,000 people is not a small anecdote. It’s a large enough signal to make dedicated addiction trials a clear priority.
Metabolic Relief Is Probably Part of the Story
Semaglutide doesn’t have to act like an antidepressant to improve psychiatric outcomes. Effective treatment of obesity, diabetes, and metabolic syndrome changes a remarkable number of mood-relevant variables at once: sleep apnea, chronic pain, inflammatory burden, glucose variability, energy, mobility, and self-perception.
Poor sleep worsens anxiety regulation and emotional control. Insulin resistance and systemic inflammation overlap with fatigue and low mood. Chronic pain and limited mobility shrink social contact and reward. Any one of those pathways could feed a meaningful reduction in severe psychiatric events even if semaglutide never touches a serotonin receptor.
The registry can’t separate these routes. It shows that severe psychiatric events were less common during GLP-1 treatment periods — not which biological mechanism was responsible. That distinction matters for how clinicians should think about these drugs.
This Study Answers a Safety Question, Not an Efficacy Question
The study was designed around psychiatric safety: do GLP-1 drugs make things worse for people with depression or anxiety? The answer is clearly no — things improved by several measures. But that doesn’t mean semaglutide is a psychiatric treatment.
Hospital care and sickness absence are severe-event markers, not symptom scales. They capture episodes bad enough to reach the health system or disrupt work. A drug could reduce severe crises without touching baseline mood scores. A reduction in suicidal behavior (also reported in this paper) is clinically significant, but it reflects different biology than remission from a depressive episode.
The practical implication is reassurance for prescribers: GLP-1 drugs appear safe to use in people with depression and anxiety, and the registry points toward possible psychiatric benefit rather than harm. But “fewer hospitalizations” is not the same as “treats depression.” That question needs symptom-level randomized trials.
The Next Trials Should Measure Mechanisms, Not Just Outcomes
The most important follow-up research would separate the routes. A well-designed trial could track weight change, metabolic markers, inflammatory biomarkers, sleep quality, craving scores, and depressive symptoms in the same people — then use mediation analysis to ask how much of the psychiatric improvement traveled through metabolic repair versus direct central GLP-1 effects.
That distinction matters for patient selection. If the benefit is mostly indirect (metabolic improvement → better sleep → improved mood), then the clearest psychiatric benefit should appear in people with significant metabolic comorbidity. If there’s a direct reward-circuit or anti-craving effect, the benefit might extend to people regardless of their metabolic profile.
For now, the data support a modest but meaningful reframe: GLP-1 drugs aren’t just diabetes and weight-loss medications for people with psychiatric disorders. They appear to reduce severe psychiatric events. The mechanism is worth finding.
Citation: Taipale et al. Association between GLP-1 receptor agonist use and worsening mental illness in people with depression and anxiety in Sweden: a national cohort study. The Lancet Psychiatry. 2026. DOI: 10.1016/S2215-0366(26)00014-3
Study Design: Swedish national registry cohort study; within-person comparison of GLP-1 on versus off periods, 2009–2022.
Sample Size: ~100,000 people with depression or anxiety; >20,000 GLP-1 users.
Key Statistic: During semaglutide treatment periods: psychiatric hospital visits and sickness absence 42% lower; depression outcomes 44% lower; substance-use outcomes 47% lower.