TL;DR: A 2026 mouse preprint in bioRxiv found that psilocybin reduced firing in somatostatin (SST) interneurons, inhibitory cortical cells that gate dendritic input, while increasing parvalbumin (PV) interneuron firing in the medial frontal cortex.
Key Findings
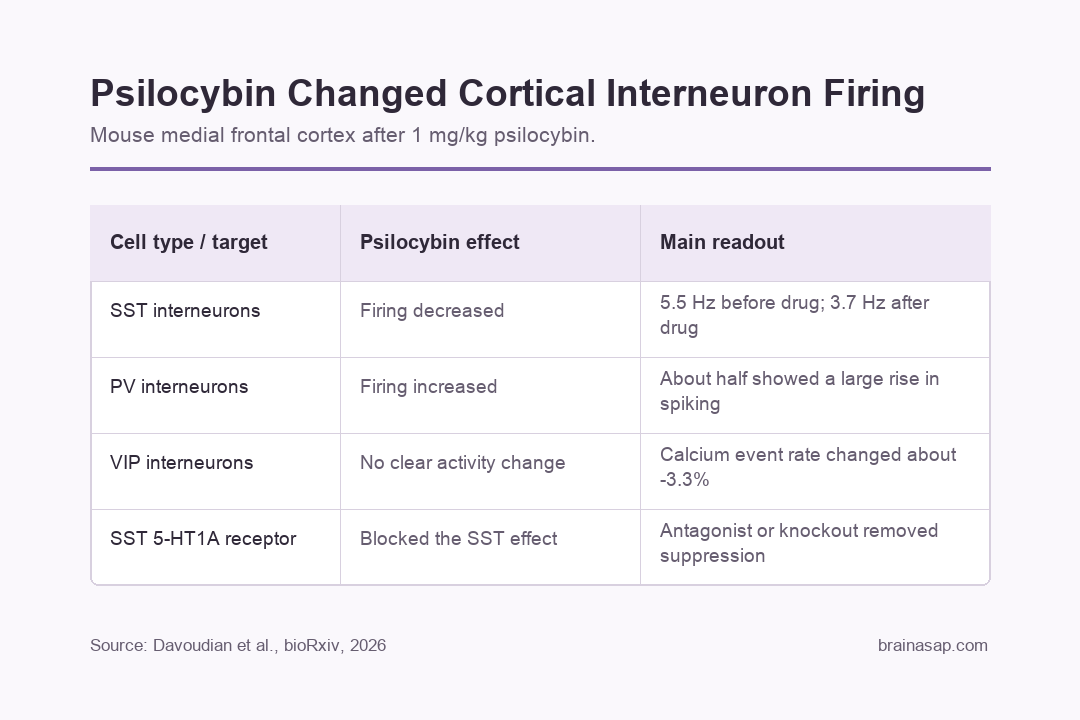
- SST firing decreased: Opto-tagged SST interneurons fell from 5.5 to 3.7 Hz after psilocybin, while saline did not produce the same drop.
- PV firing increased: PV interneurons, another major inhibitory cell class, showed higher spiking after psilocybin, with about half of recorded PV cells showing a large rise.
- VIP cells did not explain the SST drop: VIP interneuron calcium event rates changed by about -3.3% after psilocybin, close to the saline change of -2.8%.
- 5-HT1A signaling mattered: Blocking 5-HT1A receptors before psilocybin prevented the usual SST firing reduction.
- Behavioral effects depended on SST 5-HT1A receptors: Deleting 5-HT1A receptors from SST cells removed psilocybin’s longer-term effects in fear-extinction and tail-suspension assays, but not the acute head-twitch response.
Source: bioRxiv (2026) | Davoudian et al.
Psilocybin is usually discussed through serotonin 5-HT2A receptors and excitatory cortical neurons. This mouse study focused instead on inhibitory neurons, because GABAergic interneurons also carry serotonin receptors and shape how cortical circuits process input.
Psilocybin Suppressed SST Interneurons in Mouse Frontal Cortex
Researchers recorded from genetically identified interneurons in the mouse medial frontal cortex using Neuropixels probes and opto-tagging. Opto-tagging means the cells were made light-responsive, letting researchers identify which recorded units belonged to a specific interneuron class.
SST interneurons fired less after psilocybin. Their average firing rate fell from 5.5 Hz before drug to 3.7 Hz after drug, while saline did not produce a significant reduction.
That reduction was not uniform across every recorded cell. Using a large z-score drop as the threshold, 18.5% of SST interneurons showed a substantial firing decrease after psilocybin, compared with none after saline.
SST cells inhibit dendrites, the branch-like receiving zones of pyramidal neurons. Reducing SST firing can therefore loosen dendritic inhibition, making it easier for incoming signals to influence cortical neurons.
PV Interneurons Increased Firing After Psilocybin
PV interneurons showed the opposite direction. These cells mostly target the cell body region of neurons and help control spike timing, so their role is different from the dendrite-focused gating done by SST cells.
After psilocybin, PV interneuron firing increased about 15 minutes after injection. About half of the recorded PV cells showed a substantial rise in spiking.
The combination points to a more specific circuit effect than simple excitation or inhibition:
- Lower SST activity: Less dendritic inhibition may allow stronger input processing at pyramidal-cell dendrites.
- Higher PV activity: More perisomatic inhibition may constrain excessive output spiking near the cell body.
- Cell-type specificity: Psilocybin changed inhibitory-cell populations in different directions instead of globally silencing or activating all interneurons.
VIP Interneurons Did Not Drive the SST Effect
One plausible mechanism was a disinhibitory chain: psilocybin might activate VIP interneurons, and VIP cells might then inhibit SST cells. The imaging data did not support that explanation.
In awake, head-fixed mice, VIP interneuron calcium event rates changed by about -3.3% after psilocybin and -2.8% after saline. That near-match made VIP activation unlikely to be the reason SST firing fell.
Researchers also measured SST and PV calcium signals in superficial cortical layers. SST calcium events decreased after psilocybin, matching the electrophysiology result, while PV calcium imaging did not show the same clear increase seen in deeper Neuropixels recordings.
5-HT1A Receptors on SST Cells Carried the Suppression
SST interneurons expressed 5-HT1A receptors, a serotonin receptor type that can reduce neuronal excitability. Researchers tested that pathway in two ways.
- Pharmacological blockade: Pretreatment with WAY-100635, a 5-HT1A receptor antagonist, prevented psilocybin from reducing SST firing.
- Conditional knockout: Mice with reduced 5-HT1A receptor expression in SST interneurons also lost the usual psilocybin-induced SST suppression.
- Receptor specificity: These experiments pointed to 5-HT1A activity at SST cells rather than VIP-cell disinhibition as the main route for the SST firing change.
The knockout was not complete: Htr1a transcripts appeared in 44% of SST cells in controls and 14% of SST cells in conditional knockout mice. Even with that partial deletion, the psilocybin effect on SST firing was removed.
SST 5-HT1A Deletion Removed Longer-Term Stress-Behavior Effects
The behavioral experiments asked whether the SST 5-HT1A pathway mattered beyond acute circuit physiology. Researchers tested fear extinction, tail suspension, and the head-twitch response.
In control mice, psilocybin reduced freezing during fear extinction and extinction retention. Those effects were absent when 5-HT1A receptors were deleted from SST interneurons, with genotype-by-treatment interactions of P = 0.002 and P = 0.001.
Psilocybin also reduced immobility time in the tail suspension test in control mice, but not in SST-1A conditional knockout mice. The genotype-by-treatment interaction for that assay was P = 0.035.
The acute head-twitch response was different. Removing 5-HT1A receptors from SST cells did not block the head-twitch response, which fits the idea that acute psychedelic-like behavior and longer-term stress-related behavior depend on partly separable mechanisms.
The Mouse Data Narrow Psilocybin’s Cortical Mechanism
This is a mechanism study, not a clinical efficacy study. It does not show that SST interneurons explain antidepressant effects in humans, and it does not test patients with depression or anxiety.
The useful biological point is narrower: psilocybin reshaped inhibitory cortical control in mice, and the direction depended on interneuron subtype. SST cells were suppressed through a 5-HT1A-dependent pathway, while PV cells increased firing.
For human translation, the next step is to ask whether comparable inhibitory-cell effects appear in human cortical tissue, brain-wave recordings, brain-imaging measures, or other circuit-level readouts after psilocybin. The mouse work gives researchers a specific circuit target instead of a broad psychedelic-mechanism label.
Citation: DOI: 10.64898/2026.04.16.718963. Davoudian et al. Psilocybin reshapes cortical inhibition through selective interneuron recruitment. bioRxiv. 2026.
Study Design: Mouse mechanism study using Neuropixels recordings, calcium imaging, receptor blockade, conditional knockout, and behavioral assays.
Sample/Model: Genetically defined mouse cortical interneuron recordings and stress-related behavioral tests.
Key Statistic: SST interneuron firing fell from 5.5 to 3.7 Hz after psilocybin, and SST-specific 5-HT1A deletion removed psilocybin’s longer-term fear-extinction and tail-suspension effects.
Caveat: The preprint is a mouse mechanism study and has not been certified by peer review.