TL;DR: A 2026 medRxiv preprint reported that ventral capsule deep brain stimulation (DBS), an implanted brain-stimulation treatment, reduced severe treatment-resistant obsessive-compulsive disorder (OCD) symptoms by 22 Yale-Brown Obsessive-Compulsive Scale (Y-BOCS) points on average in 10 participants.
Key Findings
- 10 severe OCD patients: Researchers implanted bilateral ventral internal capsule DBS leads in participants with intractable obsessive-compulsive disorder.
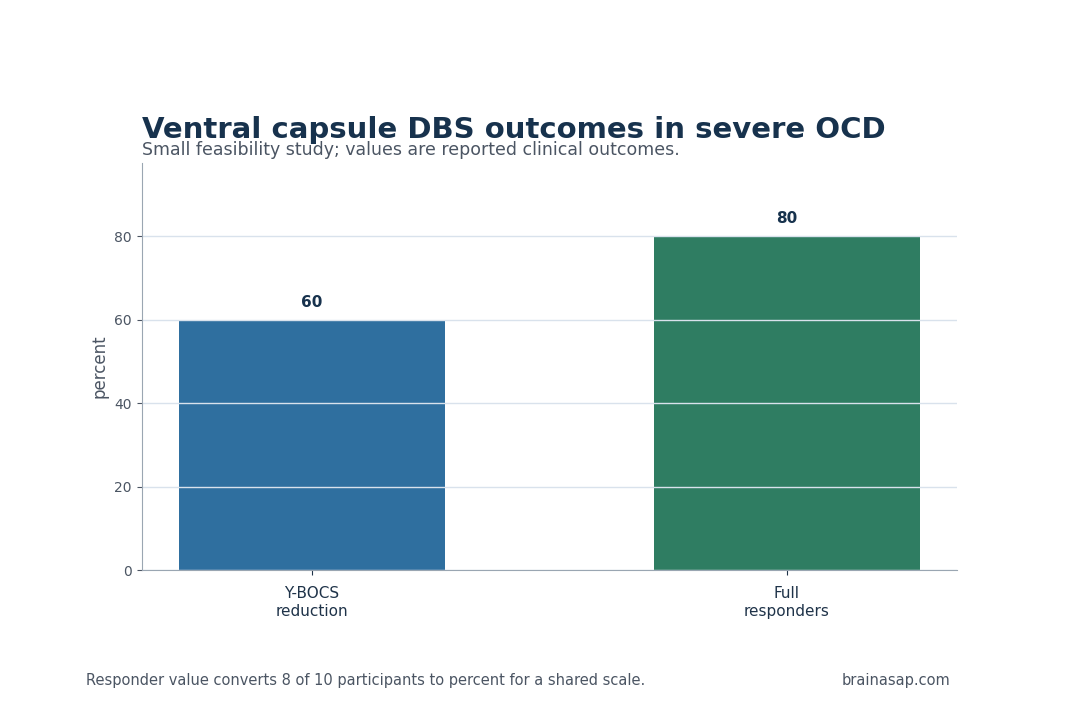
- 60% mean symptom reduction: End-of-study Y-BOCS scores, the standard OCD severity scale, fell by 22 points on average.
- 8 full responders: Eight of 10 participants met the study definition of full response, set at more than 35% Y-BOCS reduction.
- DBS discontinuation tested dependence: A double-blind discontinuation phase compared true stimulation with sham conditions after programming and psychotherapy phases.
- At-home neural sensing was feasible: The bidirectional devices recorded neural data in the clinic and at home, which can help future DBS programming move beyond trial and error.
Source: medRxiv (2026) | Agarwal et al.
Ventral Capsule DBS Targeted Severe Treatment-Resistant OCD
Obsessive-compulsive disorder can become disabling when intrusive fears and repetitive behaviors continue despite medication and specialized psychotherapy. The group in this feasibility study sat at that hard end of the clinical spectrum: severe, treatment-resistant OCD requiring an implanted neuromodulation approach rather than another routine medication trial.
Researchers placed bilateral leads in the ventral internal capsule, a white-matter region often targeted when DBS is used for psychiatric illness.
The first aim was clinical: determine whether stimulation could reduce OCD severity enough to justify the risk and complexity of the procedure. A second aim was technical: determine whether sensing-capable hardware could record useful neural data during real life, not only during brief clinic sessions.
That distinction is practical. Standard DBS programming can take months because clinicians change settings, wait for symptoms to move, and adjust again.
A device that records brain activity during daily life would eventually give clinicians a more objective readout of therapeutic state, although this early study was not designed to prove a closed-loop treatment algorithm.
Y-BOCS Scores Fell by 22 Points After DBS Programming
The main clinical number was large for a population this ill. By the end of follow-up, mean Y-BOCS score had fallen by 22 points, or about 60%.
The Yale-Brown Obsessive-Compulsive Scale is the standard clinician-rated measure of OCD severity, so that change represents a substantial shift in the symptom burden the treatment was trying to reduce.
Response also appeared across individuals, not only in an average pulled upward by a small number of exceptional cases. Eight of the 10 participants met the full-response threshold of more than 35% reduction in Y-BOCS score.
All participants also had lower depression severity, a clinically relevant secondary observation because severe OCD often travels with mood symptoms, distress, and impaired daily functioning.
- Core outcome: OCD severity was measured with Y-BOCS before and after staged DBS programming and follow-up.
- Responder threshold: Full response required more than 35% symptom reduction, not only a small numerical change.
- Mood symptoms: Depression severity decreased across the study, though the preprint summary does not make depression the primary endpoint.
The study should not be read as evidence that DBS belongs early in OCD care. The population was selected for severe, refractory illness.
For that group, however, the magnitude of the Y-BOCS change supports ventral capsule DBS as a serious treatment candidate rather than a last-ditch idea without measurable clinical movement.
Sensing-Enabled DBS Could Improve OCD Programming
The technical part of the study has the longest clinical runway. Participants received investigational bidirectional DBS devices, meaning the same system could stimulate and record neural signals.
The second half of the cohort also received strip electrodes over bilateral orbitofrontal cortex for recording only, expanding the measurement space beyond the implanted stimulation target.
OCD DBS programming is still heavily empirical. Clinicians choose settings, observe symptom change, watch side effects, and revise.
If a neural biomarker can indicate when stimulation is moving a patient toward therapeutic response, future programming can become faster and less dependent on delayed symptom reports.
Researchers showed that neural data could be recorded both at home and in the clinic. Psychiatric symptoms fluctuate with context, stress, sleep, and exposure to triggers.
Clinic-only recordings can miss the states that drive impairment during normal life.
ERP Psychotherapy and Sham Discontinuation Strengthened the Design
DBS response can be hard to interpret when expectation, programming, and psychotherapy all overlap. Participants first completed scheduled DBS programming visits.
They then completed exposure and response prevention (ERP), the behavioral therapy that asks patients to face feared cues without performing compulsions.
After that came a double-blind discontinuation phase comparing active stimulation with sham conditions.
The design does not remove every interpretive challenge, but it does more than an open-label case series. The discontinuation phase tested whether patients still preferred stimulation when blinded conditions were introduced.
The preprint reports that all participants elected to resume stimulation after discontinuation, which is consistent with perceived clinical benefit.
- Phase 1: Stimulation parameters were adjusted through scheduled programming visits.
- Phase 2: Participants received ERP psychotherapy after DBS programming.
- Phase 3: A double-blind discontinuation compared true and sham stimulation.
- Phase 4: Open-label follow-up tracked later clinical status.
That staged structure is useful because severe OCD treatment often requires combined approaches.
DBS can reduce the rigidity or distress that keeps ERP from working, while ERP can help patients convert improved brain-state control into changed behavior. The preprint supports that combined-treatment logic without proving which component contributed how much.
The sequencing also reflects how severe OCD is treated in practice. A device can change the physiological conditions under which therapy occurs, but patients still need repeated exposure to feared cues and practice resisting compulsions.
Measuring the clinical course across both stimulation and ERP therefore gives a more realistic view of care than testing stimulation in isolation.
Small OCD DBS Studies Need Careful Replication
The main caution is sample size. A 10-person cohort is enough for an early feasibility study, not enough for a general treatment rule.
Surgical selection also matters: people who qualify for psychiatric DBS often differ from the broader OCD population in illness duration, prior treatment exposure, impairment, and willingness to accept device-related risk.
There are also conflicts and device-industry links disclosed in the source material, including donated DBS hardware and multiple author relationships with device or mental health companies.
Those disclosures do not negate the clinical data, but they raise the value of independent replication and transparent adverse-event reporting in larger trials.
Adverse-event detail will be especially important in the next stage. Psychiatric DBS requires neurosurgery, implanted hardware, repeated programming, and long-term device management.
A response rate can look strong while the treatment remains appropriate only for a narrow group if surgical risk, device burden, or mood-related side effects are substantial.
Durability is another open endpoint. OCD symptoms can fluctuate with stress and exposure patterns, so longer follow-up should show whether benefit persists after programming stabilizes and psychotherapy sessions end.
The best current interpretation is specific: in a small, severe, treatment-resistant OCD cohort, ventral capsule DBS produced large Y-BOCS reductions and showed that sensing-capable devices can collect neural recordings during everyday life.
The next step is to learn whether those recordings can guide programming, predict relapse, or support closed-loop stimulation in a way that improves outcomes beyond expert manual adjustment.
Citation: DOI: 10.64898/2026.04.24.26351318; Agarwal et al.; Sensing-enabled ventral capsule DBS in intractable obsessive-compulsive disorder; medRxiv; 2026.
Study Design: Early feasibility study with staged DBS programming, ERP psychotherapy, blinded discontinuation, and open-label follow-up.
Sample Size: 10 participants with severe, treatment-resistant OCD.
Key Statistic: Mean Y-BOCS score decreased by 22 points, or 60%, and 8 of 10 participants met full-response criteria.
Caveat: Small surgical feasibility cohort; larger independent trials are needed before broad clinical use.