TL;DR: A 2026 medRxiv preprint found that white matter hyperintensity (WMH) burden, the amount of MRI-visible white-matter injury, was greater and progressed faster in amyotrophic lateral sclerosis (ALS), with progression tied to worse motor and cognitive scores.
Key Findings
- 369-person MRI sample: Researchers analyzed 204 ALS patients and 165 healthy controls from the Canadian ALS Neuroimaging Consortium.
- 35.7% higher baseline WMH burden: ALS patients had greater white matter hyperintensity burden at baseline than matched controls.
- 0.9 cc more one-year progression: ALS patients accumulated more WMH volume over one year than controls.
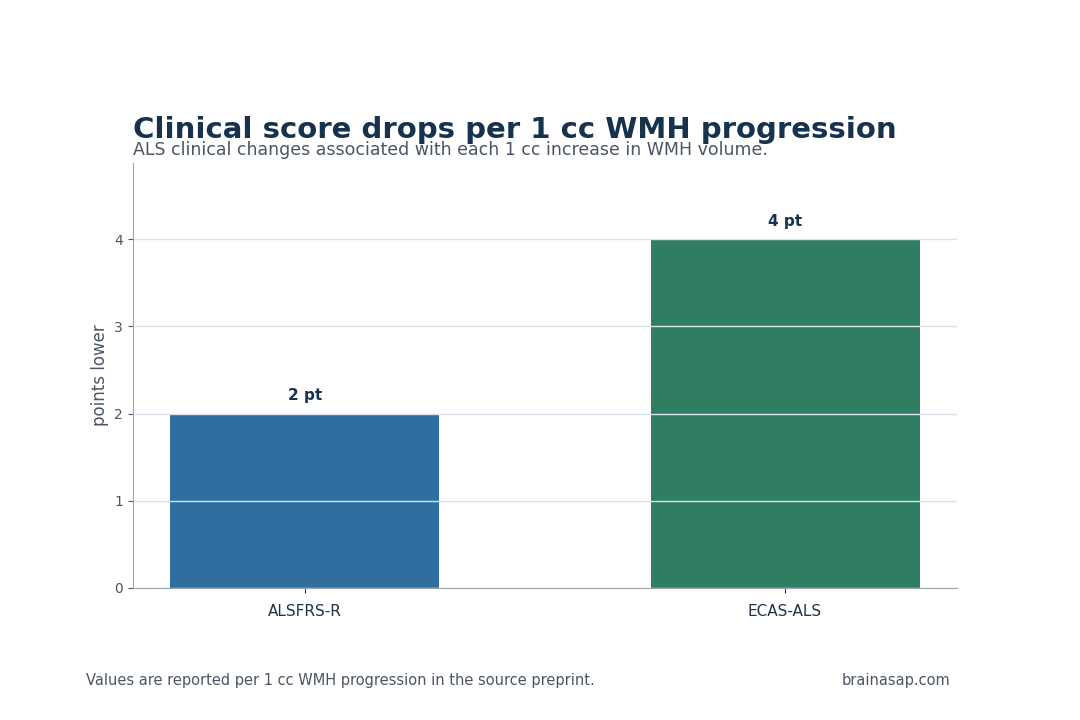
- Motor and cognitive scores declined with WMHs: Each 1 cc of WMH progression aligned with a 2-point ALSFRS-R drop and a 4-point ECAS-ALS drop.
- Short-survival ALS had faster WMH progression: The short-survival subgroup had 0.690 cc per year faster WMH progression than the long-survival subgroup.
- Riluzole and edaravone groups progressed slower: Patients taking these medications had slower WMH progression than non-treatment groups.
Source: medRxiv (2026) | CALSNIC research team
ALS Patients Had More White Matter Hyperintensity Burden
Amyotrophic lateral sclerosis is best known as a motor neuron disease, but clinical decline can also involve cognition, behavior, frontotemporal-network changes, and broader brain vulnerability. That wider presentation gives MRI markers a reason to be tested alongside motor and survival measures.
Researchers used the Canadian ALS Neuroimaging Consortium (CALSNIC), a multicenter dataset with MRI and clinical assessment across three time points over one year.
After exclusions, the analysis included 204 ALS patients and 165 controls. ALS patients had 35.7% greater WMH burden at baseline than age- and sex-matched controls.
That baseline difference was only one readout. Over one year, ALS patients had 0.9 cubic centimeters more WMH progression than controls.
In an ALS context, a changing MRI burden is more clinically informative than a static difference because disease progression is the main treatment and trial-design problem.
WMHs are not unique to ALS, which is why the longitudinal design is important. A single scan can capture age-related vascular burden or prior injury.
Repeated scans can show whether white-matter change is moving alongside the disease course. Progression rate is therefore a more informative candidate marker than burden alone.
Each 1 cc WMH Increase Aligned With ALSFRS-R and ECAS-ALS Declines
The clinical associations were direct. For every 1 cc of WMH progression, ALS patients had a 2-point drop in ALSFRS-R and a 4-point drop in ECAS-ALS.
ALSFRS-R is the revised ALS Functional Rating Scale, used to track motor and daily-function decline. ECAS-ALS is the Edinburgh Cognitive and Behavioural ALS Screen, adapted for cognitive and behavioral assessment in ALS.
Those paired outcomes widen the interpretation beyond motor neurons alone.
A marker associated with ALSFRS-R can help forecast daily-function decline, while an ECAS-ALS association suggests relevance to the cognitive and behavioral side of the disease. That dual link is why WMH progression deserves attention in multidisciplinary ALS clinics.
The cognitive association is especially relevant because ALS care can miss subtle executive and behavioral changes when motor decline dominates the visit.
A standard MRI marker that rises with ECAS-ALS decline can help identify patients who need more cognitive screening, caregiver support, or frontotemporal-dementia evaluation.
It can also improve trial interpretation. If cognitive screening worsens while motor scores are stable, or the reverse occurs, WMH progression can help show whether the brain MRI course is tracking a broader neurodegenerative burden.
- Motor function: Each 1 cc WMH progression aligned with a 2-point ALSFRS-R decline.
- Cognitive-behavioral screen: Each 1 cc WMH progression aligned with a 4-point ECAS-ALS decline.
- Progression marker: WMH change over one year carried stronger clinical meaning than a single image alone.
The source does not prove that WMHs drive ALS progression. WMHs can reflect vascular injury, neuroinflammatory stress, shared vulnerability, treatment differences, or disease severity.
Still, the repeated links to clinical scores make WMH progression a candidate MRI biomarker worth testing in prognosis models.
Short-Survival ALS Had Faster WMH Progression
Survival subgroup analysis extended the clinical relevance. The short-survival group, with 51 patients, had faster WMH progression than the long-survival group, with 75 patients. The reported difference was 0.690 cc per year.
That finding suggests WMH accumulation tracks a more aggressive ALS trajectory.
Survival associations in ALS require careful handling because many factors influence survival, including age at onset, respiratory involvement, bulbar symptoms, nutrition, genetics, access to multidisciplinary care, and treatment exposure.
WMHs are unlikely to replace those variables. Their potential value is additive: a routinely visible MRI feature can help refine risk estimates when combined with clinical data.
Riluzole and Edaravone Groups Had Slower WMH Progression
The medication analysis is one of the most provocative parts of the preprint. Patients taking edaravone and riluzole had slower WMH progression than those who were not taking those medications.
The reported differences were 0.764 cc per year for edaravone and 0.924 cc per year for riluzole, compared with non-treatment groups.
That observation should be interpreted cautiously. Medication use in observational ALS data is not randomized.
Treated and untreated patients can differ in disease stage, access to care, clinician behavior, survival time, contraindications, or willingness to pursue therapies.
The association still raises a measurable imaging endpoint that could be tested prospectively.
A prospective trial would ask a simpler endpoint: when treatment exposure is assigned or carefully matched, does WMH progression slow in parallel with clinical decline?
If the answer is yes, WMH change can help explain treatment effects or identify patients whose brain MRI course differs from their motor-score course.
- Edaravone group: Slower WMH progression than participants not taking edaravone.
- Riluzole group: Slower WMH progression than participants not taking riluzole.
- Trial-design idea: WMH progression can be evaluated as an MRI endpoint in future ALS treatment studies.
WMHs Could Join ALS Prognosis and Trial Design
The study’s strongest practical use is not diagnosis. ALS diagnosis depends on clinical, electrodiagnostic, and exclusion criteria.
WMHs are common in aging and many neurological conditions, so they are not specific enough to identify ALS on their own.
The stronger role is prognosis and trial enrichment. If WMH burden and progression repeatedly align with disease severity, functional decline, cognitive screening, survival, and treatment exposure, they can help stratify patients in research.
A trial might use WMH burden to balance groups, identify vascular-brain comorbidity, or test whether a therapy slows both clinical decline and white-matter injury.
The main limitation is observational design. CALSNIC provides valuable multicenter longitudinal imaging, but associations still need replication and causal testing.
Future work should clarify whether WMHs reflect ALS-specific mechanisms, vascular comorbidity, aging-related vulnerability, medication selection, or several overlapping processes.
For now, the source gives a testable hypothesis: ALS progression can be partly visible in the changing white matter burden that standard MRI already captures.
The measurement is also practical because WMHs are already visible on standard brain MRI sequences. A future prognostic model would not need an exotic assay if validated WMH processing can be added to scans many neurological centers already collect.
The remaining challenge is specificity. WMHs can reflect vascular disease and aging, so any ALS model would need to adjust for vascular risk factors rather than assuming every lesion belongs to ALS biology.
Citation: DOI: 10.64898/2026.04.28.26351973; White matter hyperintensities as biomarkers in ALS severity progression survival and medication response; medRxiv; 2026.
Study Design: Retrospective analysis of prospectively collected multicenter longitudinal MRI and ALS clinical data.
Sample Size: 204 ALS patients and 165 controls.
Key Statistic: Each 1 cc WMH progression aligned with a 2-point ALSFRS-R drop and a 4-point ECAS-ALS drop.
Caveat: Observational medication and survival analyses cannot prove that WMH change causes ALS progression or treatment response.