TL;DR: A 2026 systematic review and exploratory network meta-analysis of 5 randomized trials in Neurological Sciences found dose-specific ON-medication motor improvements, measured while usual Parkinson’s drugs were active, for some GLP-1 receptor agonists, glucose-related metabolic hormone drugs, in Parkinson’s disease, but broader motor, non-motor, and quality-of-life evidence remained limited.
Key Findings
- 5 randomized trials: The review included 708 people with Parkinson’s disease across trials of exenatide, lixisenatide, and NLY01.
- ON/OFF medication states: ON-state means motor testing while usual Parkinson’s drugs are active; OFF-state means testing after dopaminergic medication has been withheld.
- Class-level result was not significant: Pairwise meta-analysis showed no significant overall MDS-UPDRS Part III ON-state motor improvement (MD -2.00; 95% CI, -5.46 to 1.46).
- Exenatide 20 micrograms/day ranked strongest: Network meta-analysis estimated a significant ON-state motor improvement versus control (MD -9.80; 95% CI, -14.47 to -5.13).
- Lixisenatide 20 micrograms/day also improved ON-state motor scores: The network estimate was MD -3.08 (95% CI, -5.31 to -0.85).
- OFF-state and daily-living domains were neutral: MDS-UPDRS Part III OFF state and Parts I, II, and IV did not show consistent significant benefit.
- GI adverse events were more common: GLP-1 receptor agonists increased nausea, vomiting, and constipation compared with control.
Source: Neurological Sciences (2026) | Ali et al.
GLP-1 Receptor Agonists Were Compared in Parkinson’s Disease Trials
GLP-1 receptor agonists are diabetes and obesity drugs that have attracted Parkinson’s disease interest because they may influence inflammation, insulin signaling, mitochondrial stress, and dopaminergic neuron vulnerability. The question is whether those biological clues translate into measurable clinical benefit.
This review pooled randomized controlled trials rather than animal experiments. Researchers focused on clinical scales used in Parkinson’s disease, especially the Movement Disorder Society Unified Parkinson’s Disease Rating Scale Part III, or MDS-UPDRS Part III, a motor-exam score where lower scores generally mean better motor function.
The analysis compared exenatide, lixisenatide, and NLY01 regimens. Researchers also separated medication-state tests: ON-medication testing means usual Parkinson’s drugs are active during the motor exam, while OFF-medication testing means dopaminergic medication has been withheld before assessment.
5 RCTs Included 708 Parkinson’s Disease Participants
The search covered PubMed, EMBASE, Scopus, and Web of Science through February 2026. After screening 235 records and excluding duplicates and noneligible articles, 5 randomized controlled trials met the final criteria.
Those trials included 708 participants: 396 in GLP-1 receptor agonist groups and 312 in control groups. Interventions included exenatide once weekly or daily, lixisenatide 20 micrograms/day after a brief lower-dose start, and NLY01 at 2.5 mg/week or 5 mg/week.
Researchers used both direct pairwise meta-analysis and a frequentist network meta-analysis. Pairwise analysis asks what the direct trial comparisons show; network analysis can combine direct and indirect comparisons across a connected set of treatments.
- Primary endpoint: MDS-UPDRS Part III ON-state motor score, measured while usual Parkinson’s drugs were active.
- Other motor domains: MDS-UPDRS Part III OFF state, measured after dopaminergic medication withholding, plus Part II activities of daily living and Part IV motor complications.
- Non-motor and quality-of-life domains: MDS-UPDRS Part I, Parkinson’s Disease Questionnaire-39 (PDQ-39), and Non-Motor Symptoms Scale (NMSS).
- Risk of bias: All 5 trials were judged low risk of bias across assessed Cochrane RoB 2.0 domains.
The low risk-of-bias rating strengthens confidence in the trial conduct. It does not remove the main evidence limitation: only 5 trials were available, and some dose-specific findings depended on single studies or indirect network comparisons.
Pairwise Meta-Analysis Did Not Show an Overall Motor Benefit
At the class level, GLP-1 receptor agonists did not significantly improve MDS-UPDRS Part III ON-state scores. The pooled mean difference was MD -2.00, with a 95% confidence interval from -5.46 to 1.46 and p = 0.258.
That estimate favored treatment numerically, but the confidence interval crossed zero. Heterogeneity was also substantial, with I2 = 80.5%, meaning the trials varied enough that a single class-wide effect is hard to interpret.
Subgroup analysis suggested lixisenatide improved ON-state motor scores in its trial, with MD -3.08. Exenatide showed a nonsignificant class subgroup estimate, with MD -1.83 and a confidence interval from -3.78 to 0.13.
OFF-state motor testing did not show a significant benefit. Across 3 exenatide studies and 292 participants, the pooled OFF-state MDS-UPDRS Part III estimate was MD -1.99, with a 95% confidence interval from -5.33 to 1.35.
Network Meta-Analysis Found Dose-Specific ON-State Motor Effects
The exploratory network meta-analysis produced the clearest positive results. Compared with control, exenatide 20 micrograms/day had the largest estimated ON-state MDS-UPDRS Part III improvement: MD -9.80, with a 95% confidence interval from -14.47 to -5.13.
Lixisenatide 20 micrograms/day also showed a significant ON-state motor improvement, with MD -3.08 and a 95% confidence interval from -5.31 to -0.85. NLY01 at 2.5 mg/week was statistically significant as well, though the estimated effect was smaller at MD -0.70.
Not every regimen helped. Exenatide 2 mg/week and NLY01 5 mg/week did not significantly improve ON-state Part III scores versus control in the network model.
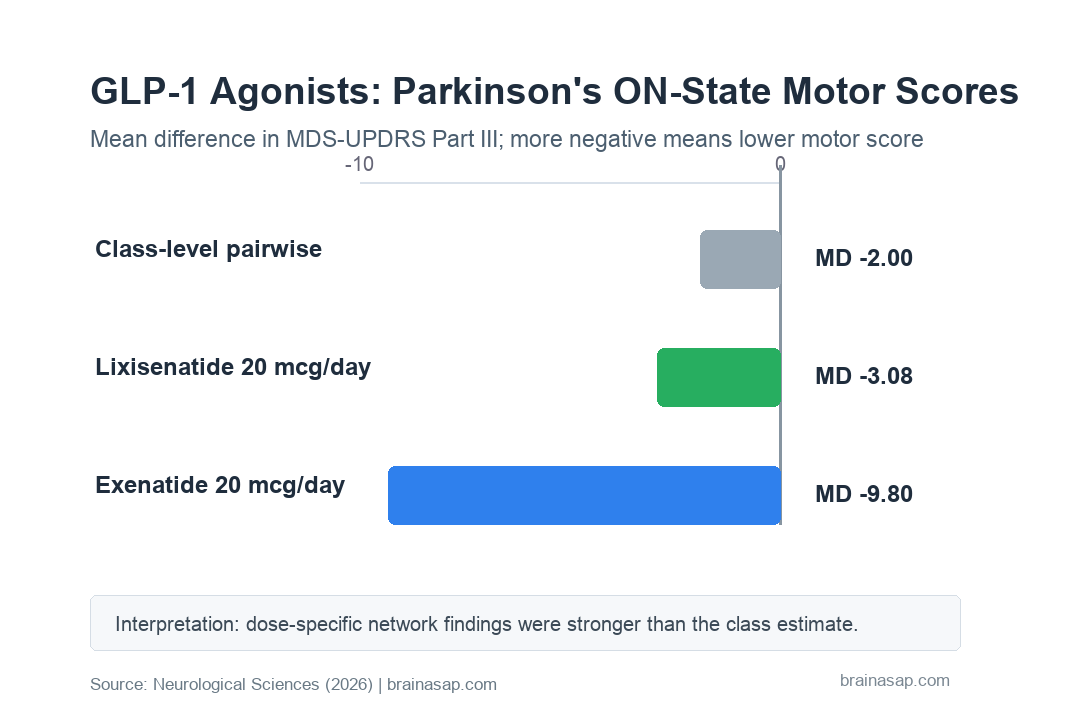
The distinction between pairwise and network results is important. The class-wide pairwise estimate was not significant, while the network model suggested that specific doses may matter more than the broad drug class label.
OFF-State, Daily Living, and Motor Complication Results Were Mostly Neutral
Broader outcomes were less convincing. MDS-UPDRS Part III OFF-state scores did not significantly improve in either pairwise or network analysis, even though the direction sometimes favored GLP-1 receptor agonists.
That weakens the disease-modification interpretation. A treatment that changes Parkinson’s disease progression would ideally show clearer benefit when ordinary dopaminergic medication is withheld, not only during ON-state testing.
MDS-UPDRS Part I, which captures non-motor experiences of daily living, showed no meaningful treatment effect. MDS-UPDRS Part II activities of daily living and Part IV motor complications were also neutral across both analytical approaches.
- Part I non-motor experiences: Estimates stayed near zero, with no significant regimen-level benefit.
- Part II daily activities: No treatment clearly improved activities of daily living.
- Part IV motor complications: No meaningful effect appeared for motor complications during studied follow-up.
- PDQ-39 quality of life: Pairwise results were neutral, while network analysis found an isolated NLY01 5 mg/week finding that needs cautious interpretation.
NMSS findings were also unstable. Pairwise analysis showed no significant effect, while network analysis gave divergent NLY01 dose results, with one dose worse and one dose better.
The authors interpreted that as imprecision rather than a reliable non-motor treatment pattern.
Gastrointestinal Side Effects Were More Frequent With GLP-1 Drugs
The safety profile looked familiar for GLP-1 receptor agonists. Gastrointestinal adverse events were more common in treated participants than controls.
Nausea was significantly increased, with a risk ratio of RR 2.09. Vomiting was also more frequent, with RR 4.53, and constipation increased with RR 1.89.
Weight loss was more common in the GLP-1 receptor agonist arms, with RR 1.81. Other adverse events, including fatigue, headache, anxiety, urinary tract infection, and administration-site disorder, did not differ significantly from controls.
For Parkinson’s disease, tolerability is not a minor detail. Nausea, vomiting, constipation, and weight loss can affect frailty, medication timing, nutrition, fall risk, and adherence in older or more vulnerable patients.
Parkinson’s GLP-1 Trials Need Larger Dose-Specific Tests
The most defensible reading is dose-specific motor potential, not broad proof of disease modification.
Exenatide 20 micrograms/day and lixisenatide 20 micrograms/day deserve attention because their ON-state motor estimates were significant and clinically visible.
Several limits should shape interpretation:
- Small evidence base: Only 5 randomized trials were available for the review.
- Heterogeneous follow-up: Treatment durations ranged from months to nearly 2 years, making long-term interpretation difficult.
- Single-study findings: Some statistically significant results came from one trial or one dose-specific network comparison.
- Limited disease-modification evidence: OFF-state motor outcomes did not show consistent significant benefit.
- Side-effect tradeoff: GI intolerance and weight loss may matter clinically in Parkinson’s disease.
The next trial question is therefore specific: which GLP-1 receptor agonist, dose, and treatment window can improve Parkinson’s motor function without creating unacceptable gastrointestinal burden?
Future studies also need cognitive, psychiatric, sleep, autonomic, and quality-of-life endpoints, not only motor scores. Parkinson’s disease disability is broader than Part III motor examination, and a repurposed metabolic drug should show benefits that patients can feel in daily function.
Citation: DOI: 10.1007/s10072-026-08929-1. Ali et al. Efficacy of GLP-1 receptor agonists in Parkinson’s disease: a systematic review and exploratory network meta-analysis of randomized controlled trials. Neurological Sciences. 2026;47:432.
Study Design: Systematic review, pairwise meta-analysis, and exploratory frequentist network meta-analysis of randomized controlled trials.
Sample Size: 5 randomized trials with 708 Parkinson’s disease participants.
Key Statistic: Network meta-analysis estimated ON-state MDS-UPDRS Part III improvement, measured while usual Parkinson’s drugs were active, with exenatide 20 micrograms/day (MD -9.80; 95% CI, -14.47 to -5.13).
Caveat: Small trial base, substantial heterogeneity in pairwise motor analysis, single-dose findings, neutral OFF-state results, and more gastrointestinal adverse events.