TL;DR: A 2026 study in JAMA Network Open tracked 1,338 older adults from the Rush Memory and Aging Project for up to 19 years using objective wrist-actigraphy and found that each additional hour of daytime napping per day was linked to ~13% higher mortality, each extra nap per day to ~7% higher mortality, and morning nappers had 30% higher mortality than afternoon nappers — though the authors stress this is correlation, not causation.
Key Findings
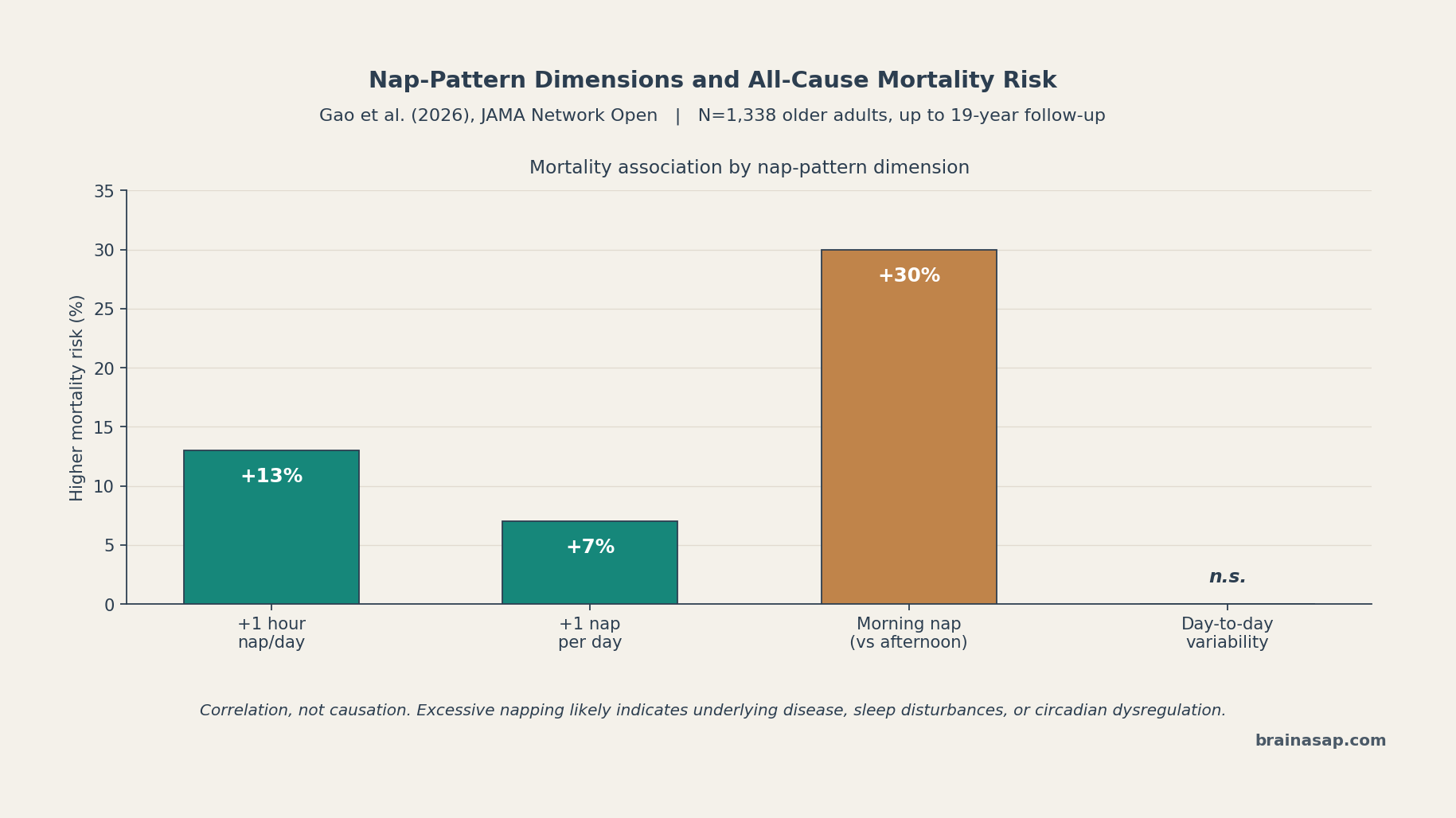
- Each extra hour of napping linked to ~13% higher mortality: Across 1,338 older adults followed for up to 19 years, each additional hour of daytime napping per day was associated with approximately 13% higher all-cause mortality.
- Each extra nap linked to ~7% higher mortality: Each additional nap per day was associated with about 7% higher mortality risk.
- Morning nappers had 30% higher mortality than afternoon nappers: Time-of-day mattered. Morning naps were specifically associated with higher mortality compared with naps later in the day.
- Day-to-day variability did not predict mortality: Irregular napping patterns — naps that came and went unpredictably from one day to the next — were not associated with increased mortality risk.
- Wrist-actigraphy data captured patterns self-report misses: Participants wore wrist activity monitors for 10 days at the initial assessment, giving objective rest-activity data rather than relying on memory-based questionnaires.
- Correlation, not causation: Lead author Chenlu Gao notes excessive napping likely indicates underlying disease, sleep disturbances, or circadian dysregulation rather than causing mortality directly. Naps may be an early warning rather than a risk factor to eliminate.
Source: JAMA Network Open (2026) | Gao et al.
Between 20% and 60% of older adults nap. Infrequent restorative naps are widely considered healthy, but excessive daytime napping in older age has been linked to neurodegeneration, cardiovascular disease, and broader morbidity in earlier studies.
Most of that earlier evidence relied on self-reported napping — participants estimating from memory how often and how long they napped — and missed the time-of-day and day-to-day variability that may matter biologically.
This study used 10 days of objective wrist-actigraphy at baseline and 19 years of mortality follow-up to test which specific nap features tracked with how long older adults lived.
1,338 Older Adults Tracked With Wrist Actigraphy and Up to 19 Years of Follow-Up
Researchers at Mass General Brigham and Rush University Medical Center analyzed the Rush Memory and Aging Project, a cohort study of older adults in northern Illinois that began in 1997.
The design:
- Cohort: 1,338 older adults from the Rush Memory and Aging Project, predominantly white.
- Baseline measurement: Beginning in 2005, participants wore wrist activity monitors for 10 days to capture continuous rest-activity data.
- Sleep features extracted: Nap length, nap frequency, time of day each nap occurred, and day-to-day variability of napping patterns.
- Outcome: All-cause mortality across the up-to-19-year follow-up window.
The wrist-actigraphy approach captures features self-report cannot. A participant who naps for 90 minutes but reports “a short rest” would be invisible on questionnaire data and visible to the actigraph.
Longer, More Frequent, and Morning Naps Tracked With Higher Mortality
The study extracted four nap-pattern dimensions and tested each against the 19-year mortality data.
The associations:
- Total nap time: Each additional hour per day of daytime napping → ~13% higher all-cause mortality.
- Nap frequency: Each additional nap per day → ~7% higher mortality risk.
- Time of day: Morning nappers had 30% higher mortality than afternoon nappers.
- Day-to-day variability: No association with mortality.
The morning-vs-afternoon split is the most novel finding. A long afternoon nap and a long morning nap of identical duration carried different prognostic information in this dataset.
Why the Morning-vs-Afternoon Split Matters Biologically
Afternoon naps are part of normal circadian biology. The post-lunch dip in alertness is driven by ordinary circadian rhythm and a brief restorative nap fits within that pattern.
Morning naps are more clinically informative because they suggest something else is wrong:
- Disrupted nighttime sleep: Someone who napped heavily in the morning likely slept poorly the night before — pointing to sleep apnea, pain, anxiety, or other nocturnal disturbance.
- Circadian dysregulation: Morning sleepiness can reflect a phase-shifted circadian clock, which becomes more common with neurodegenerative disease.
- Underlying medical illness: Cardiovascular disease, depression, and early dementia all can produce morning fatigue that pulls a person back to sleep.
None of these are caused by the morning nap itself. The nap is the visible signal of an upstream problem that also raises mortality risk.
Wrist-Actigraphy Catches What Self-Report Misses
The methodological contribution of this study is the actigraphy data.
Self-reported napping has known weaknesses:
- Recall bias: People underestimate how long they nap, especially when sleep crept up unintentionally.
- Time-of-day inaccuracy: Participants often cannot reliably report when their naps occurred.
- Variability blind spot: Day-to-day patterns are essentially invisible to retrospective questionnaires.
Wrist actigraphy captures all four dimensions continuously and objectively. The fact that the time-of-day signal showed up in the actigraphy data and not in earlier self-report studies is consistent with the prior literature having missed it for measurement reasons.
Nap Patterns May Work Better as Surveillance Than as a Behavioral Target
The clinical interpretation was direct:
“It is important to note that this is correlation not causation. Excessive napping is likely indicating underlying disease, chronic conditions, sleep disturbances, or circadian dysregulation.”
Gao’s argument is that wearable nap-pattern monitoring belongs in geriatric care as an early-warning surveillance tool, not as a behavior to be eliminated through better sleep hygiene alone.
Observational Design, White Cohort, and Confounding Limit Causal Reading
- Observational, not experimental: The associations cannot establish that naps cause mortality. The reverse and the upstream-confounder readings are both consistent with the data.
- Predominantly white northern-Illinois cohort: Generalization to other ancestry, regional, and socioeconomic groups requires separate analyses.
- Baseline-only nap measurement: The 10-day actigraphy window captured patterns at the start of follow-up; whether changing nap patterns over time predicts incremental mortality risk needs a different design.
- Underlying medical conditions not fully separated: Cardiovascular, neurodegenerative, and psychiatric conditions could each independently produce both higher mortality and the napping signature, leaving the napping pattern as a marker rather than a causal driver.
- Cause-specific mortality not reported here: The primary outcome was all-cause mortality. Whether the morning-nap signal points specifically to neurodegenerative or cardiovascular death needs cause-specific follow-up analysis.
Wearable Nap Monitoring Belongs in the Geriatric Toolkit, Not Just Sleep Clinics
The actionable framing follows from the surveillance reading:
- Routine geriatric checkups should ask about napping: A short questionnaire about nap timing and length is cheap and clinically useful as a surveillance signal.
- Wearable activity trackers can extend the surveillance: Many older adults already wear fitness watches or smartwatches. Actigraphy-style nap detection from those devices could integrate into routine clinical monitoring with no extra equipment.
- Morning naps deserve specific attention: A patient newly napping in the morning is a candidate for sleep-apnea screening, neurodegenerative-disease assessment, depression evaluation, and cardiovascular checkup — each of which has independent treatment implications.
- Don’t tell patients to stop napping: The data do not support a behavioral intervention against napping itself. The right response is to investigate why the napping shifted.
Citation: DOI: 10.1001/jamanetworkopen.2026.7938. Gao C, Cai R, Zheng X, Gaba A, Gao L, Yu L, Buchman AS, Bennett DA, Hu K, Li P. Objectively Measured Daytime Napping Patterns and All-Cause Mortality in Older Adults. JAMA Network Open. 2026.
Study Design: Prospective observational cohort study using objective wrist-actigraphy at baseline and up to 19 years of mortality follow-up.
Sample Size: 1,338 older adults from the Rush Memory and Aging Project; predominantly white northern-Illinois cohort.
Key Statistic: Each additional hour of napping per day → ~13% higher all-cause mortality; each additional nap per day → ~7%; morning nappers had 30% higher mortality vs afternoon nappers; day-to-day variability of nap patterns showed no association.
Caveat: Observational design cannot establish causation; predominantly white cohort limits generalization; baseline-only nap measurement; underlying medical conditions could independently drive both nap patterns and mortality, leaving napping as a marker rather than a target.