TL;DR: A 2026 Cell Reports mouse study found FGF21 acts through beta-klotho-expressing glutamatergic neurons in the nucleus of the solitary tract to drive protein-restriction adaptation.
Key Findings
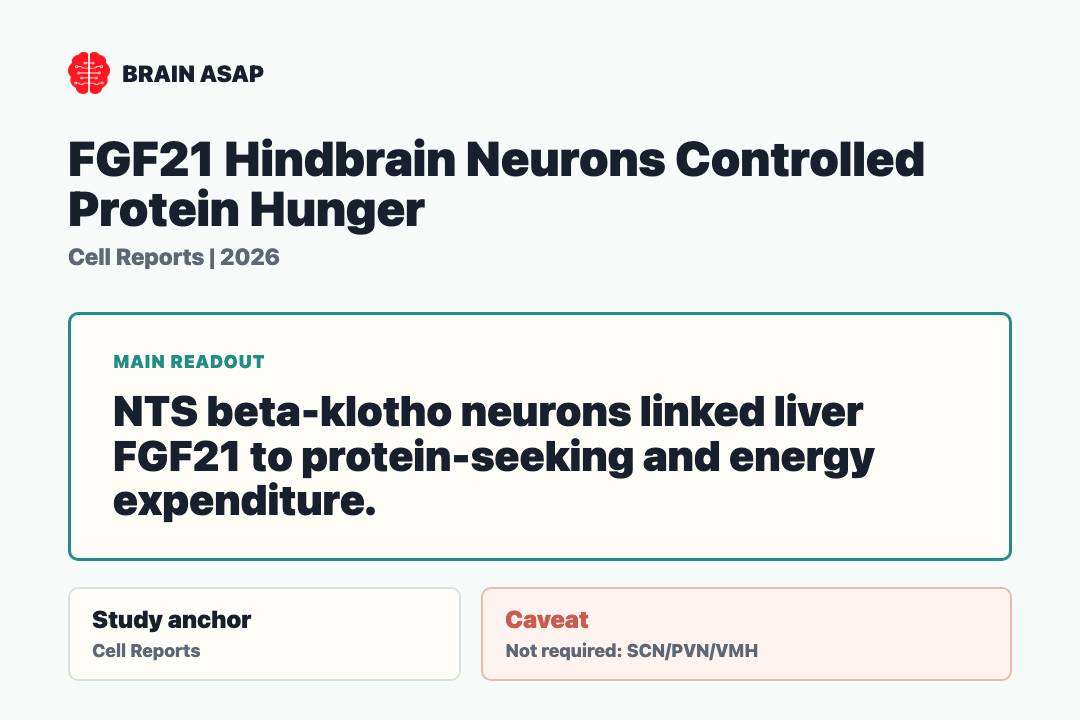
- Necessary and sufficient at one cell population: Ablating NTS-KLB neurons blocked food-intake, food-choice, and energy-expenditure changes during protein restriction. Chemogenetically activating them recreated the responses.
- The hypothalamus was not required: Direct tests showed SCN, PVN, and VMH were not needed for the FGF21-mediated responses examined — rerouting appetite control toward the brainstem.
- NTS-KLB neurons responded directly to FGF21: Beta-klotho-expressing glutamatergic neurons in the nucleus of the solitary tract translated the liver hormone into a neural command.
- Three behavioral outputs moved together: Intake, macronutrient choice, and energy expenditure all shifted as a coordinated package — not isolated dials.
- Protein hunger is a specific signal, not generic appetite: FGF21 targets a nutritional imbalance, distinct from “eat more” or “eat less.”
- Hindbrain as decision point, not relay: The NTS receives visceral information from gut and body — well placed to compare diet, liver report, and required behavior change.
Source: Cell Reports (2026) | Spann et al.
FGF21 is often described as a metabolic hormone, but hormones need circuits. This study identified a specific hindbrain population that translates a low-protein diet into actual behavior and energy use — and the location is not where most appetite research has been looking.
The Liver Signal Needed a Hindbrain Receiver
When protein intake drops, the liver releases FGF21. The hormone tells the body something specific: the diet is missing a key macronutrient, and behavior should change. The question was where that message becomes a neural command.
The answer was a discrete population of beta-klotho-expressing glutamatergic neurons in the nucleus of the solitary tract (NTS). Beta-klotho is part of the receptor machinery FGF21 uses to act on cells — finding KLB-expressing neurons in the NTS gave the authors a concrete target instead of a vague brainwide hormone effect.
Hypothalamus Wasn’t the Required Hub
Metabolic control is usually routed through hypothalamic regions like the SCN, PVN, and VMH. The authors tested those candidates directly and they were not required for FGF21-mediated responses to protein restriction. That pushes the spotlight to the hindbrain — a region sometimes treated as basic but deeply involved in appetite, visceral sensing, and energy balance.
The result revises the usual map of appetite control. The hypothalamus is central, but it is not the only place where metabolic information becomes behavior.
The NTS is well positioned for this job because it integrates visceral information, circulating signals, and feeding-related control. A liver hormone reaching a hindbrain receiver is a plausible route for changing the animal’s response to a low-protein diet — and a route that FGF21-targeting drug development should not miss.
The Necessary-and-Sufficient Pair Made the Circuit Stick
The logic was two-sided. Remove NTS-KLB neurons, and mice failed to mount normal metabolic adaptations to protein restriction. Activate them, and the responses appeared.
Necessary and sufficient at the same cell population — the strongest version of a circuit claim.
Many circuit studies stop at correlation: a population lights up during a behavior, so it becomes a candidate node. This paper went further by perturbing the cells and watching whether the adaptation survived. Together, ablation and chemogenetic activation make a much stronger case that NTS-KLB neurons sit inside the control pathway, not just along it.
The work also links outputs that are often studied separately. Intake, macronutrient choice, and energy expenditure are coordinated responses, not isolated dials — the animal is solving a nutritional imbalance, not following one appetite cue.
Protein Hunger as a Specific Signal
The study makes protein hunger feel less like willpower and more like physiology. A liver hormone reports dietary state, a hindbrain circuit receives the signal, and the animal changes intake, food choice, and energy expenditure.
That distinction matters for clinical translation. FGF21-based therapies are already being explored for metabolic disease. Understanding the route can help define endpoints beyond liver fat — including dietary behavior and metabolic rate.
Hunger is not one sensation. Protein need, energy need, gut state, reward value, and learned food preference can pull behavior in different directions. FGF21 appears to be one way the body communicates a specific nutritional problem.
That granularity also affects drug development. A therapy that changes weight without clarifying food choice, protein appetite, or energy expenditure may miss the mechanism behind durable benefit or risk.
The brainstem framing also reframes what NTS does — less a relay station, more an active decision point that compares what the animal ate, what the liver is reporting, and what behavior should change next.
Translating to Human Biology
This is mouse circuit biology. It does not say that targeting NTS-KLB neurons in humans is ready or even straightforward.
What it does is sharpen the map. If FGF21 treatments work partly through brain circuits, the next generation of metabolic therapy needs to measure not just blood chemistry but how hunger, food choice, and energy expenditure are being neurally coordinated.
Human studies would need careful endpoints. Protein preference, total intake, cravings, body composition, metabolic rate, nausea, and reward-related eating could all change in clinically important ways.
They would also need to separate protein hunger from general appetite suppression — a therapy that reduces calories while worsening protein adequacy is not the same as a therapy that helps the body correct a macronutrient imbalance.
This circuit study is useful even before translation because it gives researchers a cleaner vocabulary for asking what kind of hunger is changing. Protein appetite is a specific biological problem, and this paper finally gives it a specific circuit address. That address should make future FGF21 studies harder to keep vague for clinicians and trialists.
Citation: DOI: 10.1016/j.celrep.2026.117218. Spann et al. FGF21 signals through hindbrain neurons to alter food intake and energy expenditure during dietary protein restriction. Cell Reports. 2026.
Study Design: Mouse circuit study using Klb-Flp intersectional genetics, neuronal ablation, chemogenetic activation, and metabolic/behavioral assays.
Sample: Mice across protein-restriction, ablation, and activation experiments.
Key Result: NTS β-klotho neurons were directly activated by FGF21, required for protein-restriction adaptations, and sufficient to drive changes in food intake, food choice, and energy expenditure. Hypothalamic SCN, PVN, and VMH were not required.
Caveat: Mouse circuit biology — human translation requires endpoints that distinguish protein hunger from general appetite suppression.