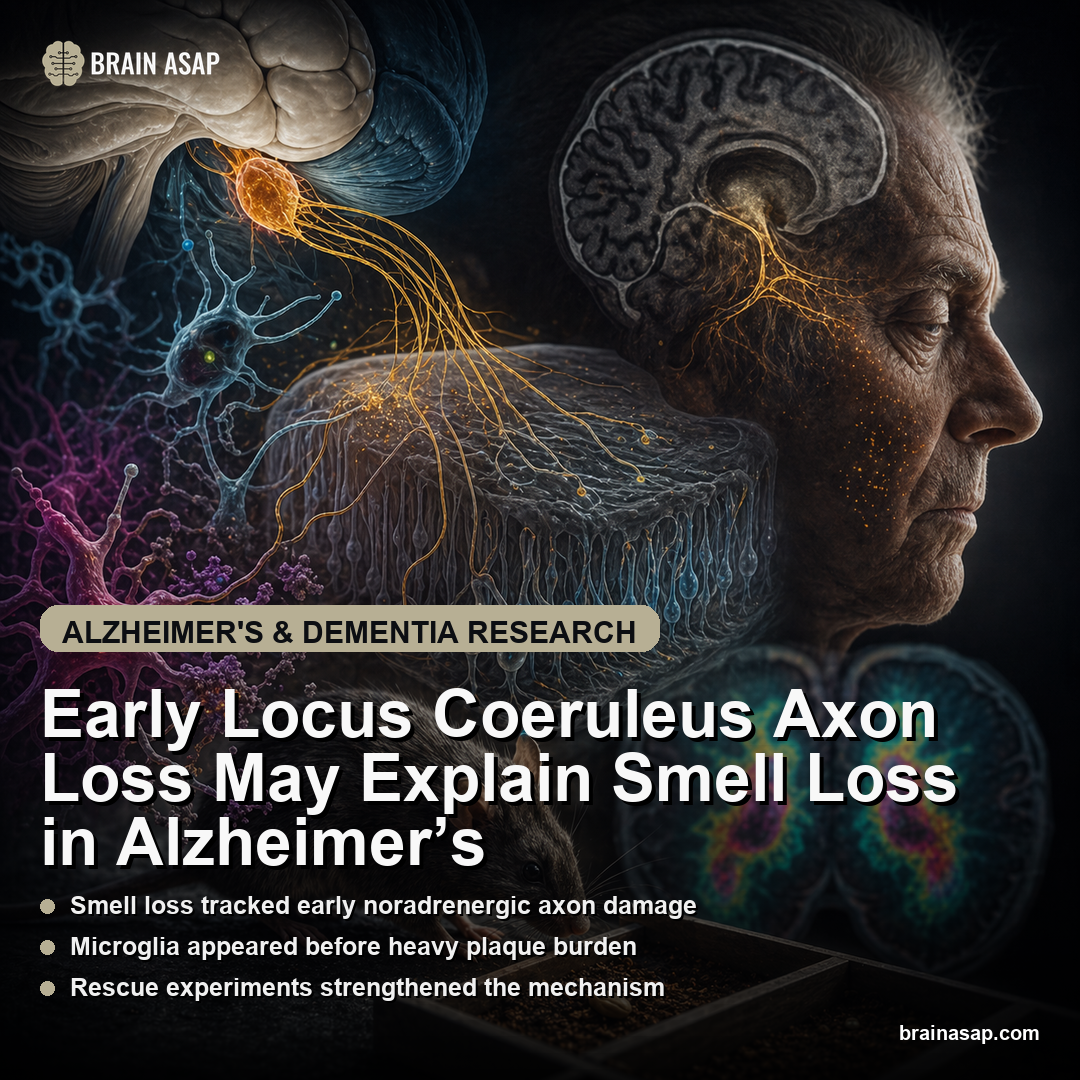
TL;DR: A 2025 Nature Communications mouse study found microglia stripped locus coeruleus axons from the olfactory bulb before major plaque buildup, offering a mechanism for early smell loss in Alzheimer’s models.
Key Findings
- TSPO knockout rescued smell: Cutting microglial phagocytosis preserved locus coeruleus axons and restored buried-food performance toward wild-type levels — the strongest causal piece.
- 60% slower buried-food at 3 months: Alzheimer’s-model mice needed roughly 60% more time to find buried food, with reduced odor-investigation behavior in a vanilla-odor test.
- Selective axon loss before heavy plaques: AppNL-G-F mice lost noradrenergic fibers in the olfactory bulb early. Cholinergic and serotonergic neurites were preserved — the damage was specific.
- 33% higher microglial phagocytosis: Olfactory-bulb microglia from AppNL-G-F mice engulfed more synaptosomes; microscopy found more LC axon material in microglial lysosomes via phosphatidylserine “eat me” signaling and MFG-E8.
- LC-restricted mutation reproduced the phenotype: Driving human AppNL-G-F only in locus coeruleus neurons still caused ~15% olfactory-bulb axon degeneration and a smell deficit.
- Human translation signal: TSPO-PET showed elevated olfactory-bulb signal in 16 prodromal AD patients vs. 14 controls; post-mortem early-AD bulbs showed reduced LC axon density.
Source: Nature Communications (2025) | Meyer et al.
Smell loss is one of the practical problems in early Alzheimer’s research: it can appear before memory failure, but the cellular explanation has often stayed vague. A useful mechanism has to connect the symptom to a specific pathway, not just to a general idea of neurodegeneration.
Early Alzheimer’s Smell Loss Needed a Clear Mechanism
Olfactory decline is one of the most repeated “early sign” claims in Alzheimer’s, yet the field has struggled to explain it cleanly. Plaques and tangles show up across many regions. Smell is processed across several stations.
Most patients with emerging smell problems get a vague warning that something neurodegenerative may be brewing — not a mechanistic account.
This paper makes the mechanism much more specific. The authors focused on the locus coeruleus, the noradrenergic brainstem hub that modulates arousal, attention, and sensory processing — including input into the olfactory bulb.
The locus coeruleus is already known to take Alzheimer’s hits early. What was missing was proof that its axons into the olfactory bulb degenerate early enough, and locally enough, to help explain smell loss before overt dementia.
Their answer was yes — and the mechanism was not passive degeneration. Microglia were actively clearing those axons.
Locus Coeruleus Axon Loss Appeared Before Olfactory Bulb Plaques
In AppNL-G-F mice, the team found early loss of noradrenergic fibers in the olfactory bulb, with the most prominent hit in the internal plexiform layer. This stood out because cholinergic and serotonergic neurites were not reduced at the same age.
The damage looked selective — not a generic collapse of every modulatory input into the region.
Behavior lined up with anatomy. At 3 months, the transgenic mice needed about 60% more time to find buried food, a classic olfactory task. In a vanilla-odor test, they showed reduced investigatory behavior.
Odor-evoked noradrenaline release in the olfactory bulb was reduced. Structural loss in the sensory circuit, impaired performance on two smell tasks, and a measurable functional deficit in the modulatory chemistry that should have been tuning the circuit.

Microglia Turned a Vulnerable Circuit Into a Removable One
This is where the paper goes past standard pathology. Bulk RNA sequencing and functional assays suggested that olfactory-bulb microglia were not just present in larger numbers — they were more phagocytic. In culture, microglia from AppNL-G-F mice showed 33% higher engulfment of labeled synaptosomes.
Microscopy pushed the mechanism further. The team found more noradrenergic axon material inside microglial lysosomes, and evidence that those axons were exposing phosphatidylserine — the classic “eat me” signal. MFG-E8 appeared to act as the adaptor linking externalized phosphatidylserine to microglial engulfment machinery.
That turns the interpretation from one of simple axon fragility to one of active removal. Hyperactive or stressed locus coeruleus axons in the olfactory bulb may be getting tagged for clearance, then physically eaten by microglia before large-scale amyloid pathology takes over the region. The story is no longer “axons died.” It is “axons were targeted.”
Rescue Experiments Made the Causal Claim Stick
Correlation alone would not have been enough. The strongest evidence comes from rescue. When the authors crossed Alzheimer’s-model mice with TSPO-knockout animals, they reduced microglial phagocytosis, preserved more locus coeruleus axons, and rescued buried-food performance at 3 months.
The inverse logic was equally clean. Restricting human AppNL-G-F expression specifically to locus coeruleus neurons still caused about 15% axon degeneration in the olfactory bulb and produced a smell deficit.
You do not need whole-brain transgenic pathology to get the phenotype. Hitting the locus coeruleus projection alone was enough. Together, the rescue and the restriction make the mechanism hard to explain as a generic plaque side effect.
Human Data Bridge Mouse to Clinic
The human arm was small but it mattered. The team used TSPO-PET in 16 prodromal Alzheimer’s patients, 16 with AD, and 14 unaffected controls.
The olfactory bulb showed elevated TSPO signal already in the prodromal group — suggesting increased microglial burden or activity before later-stage diagnosis added more signal. Post-mortem olfactory bulbs from early-AD donors showed reduced locus coeruleus axon density, matching the mouse direction.
Human olfactory testing in the prodromal group showed a trend rather than a clean deficit, and the authors are honest about the small cohort. Still, the translational bridge is unusually strong for this kind of circuit paper. TSPO-PET is a proxy for glial biology, not a direct movie of microglia eating axons, but it is the right kind of measurement for this hypothesis.
Locus Coeruleus Axon Loss Could Inform Early Alzheimer’s Detection
The clinical question is whether this circuit can be measured early enough to change care. A smell test alone is too nonspecific — allergies, infection, aging, Parkinson’s, and medications all muddy the signal.
Pairing olfactory testing with TSPO-PET, locus-coeruleus-sensitive imaging, or fluid markers of microglial activity could turn a vague early warning sign into a more biologically anchored risk marker.
This study suggests that one of Alzheimer’s earliest outward signs may be less about the nose itself and more about early loss of noradrenergic support to the olfactory bulb.
If that holds up, smell testing plus targeted imaging of this circuit could become a more biologically specific early-detection strategy — and the mechanism gives the field a real therapeutic target.
Microglial overactivity, phagocytic targeting of stressed axons, and TSPO-pathway biology are all things drug development knows how to engage. That makes this paper less a curiosity and more a roadmap.
Citation: DOI: 10.1038/s41467-025-62500-8. Meyer et al. Early Locus Coeruleus noradrenergic axon loss drives olfactory dysfunction in Alzheimer’s disease. Nature Communications. 2025;16:7338.
Study Design: Mouse circuit study with rescue experiments + small human TSPO-PET cohort + post-mortem early-AD olfactory bulb analysis.
Sample: AppNL-G-F mice, TSPO-knockout crosses, LC-restricted models; 16 prodromal AD, 16 AD, 14 controls in PET arm.
Key Result: Selective LC axon loss in olfactory bulb before heavy plaque load; 60% slower buried-food performance; TSPO knockout preserved axons and rescued behavior; human prodromal AD showed elevated olfactory-bulb TSPO-PET signal.
Caveat: Small human cohort; TSPO-PET is a glial biology proxy, not a direct microglia readout.