TL;DR: A spinal-fluid seed test caught 19 of 20 Lewy body dementia patients in a real memory-clinic cohort — and revealed hidden Lewy-body biology in 15.8% of patients carrying an Alzheimer’s diagnosis. The “Alzheimer’s” label was often correct but incomplete.
Key Findings
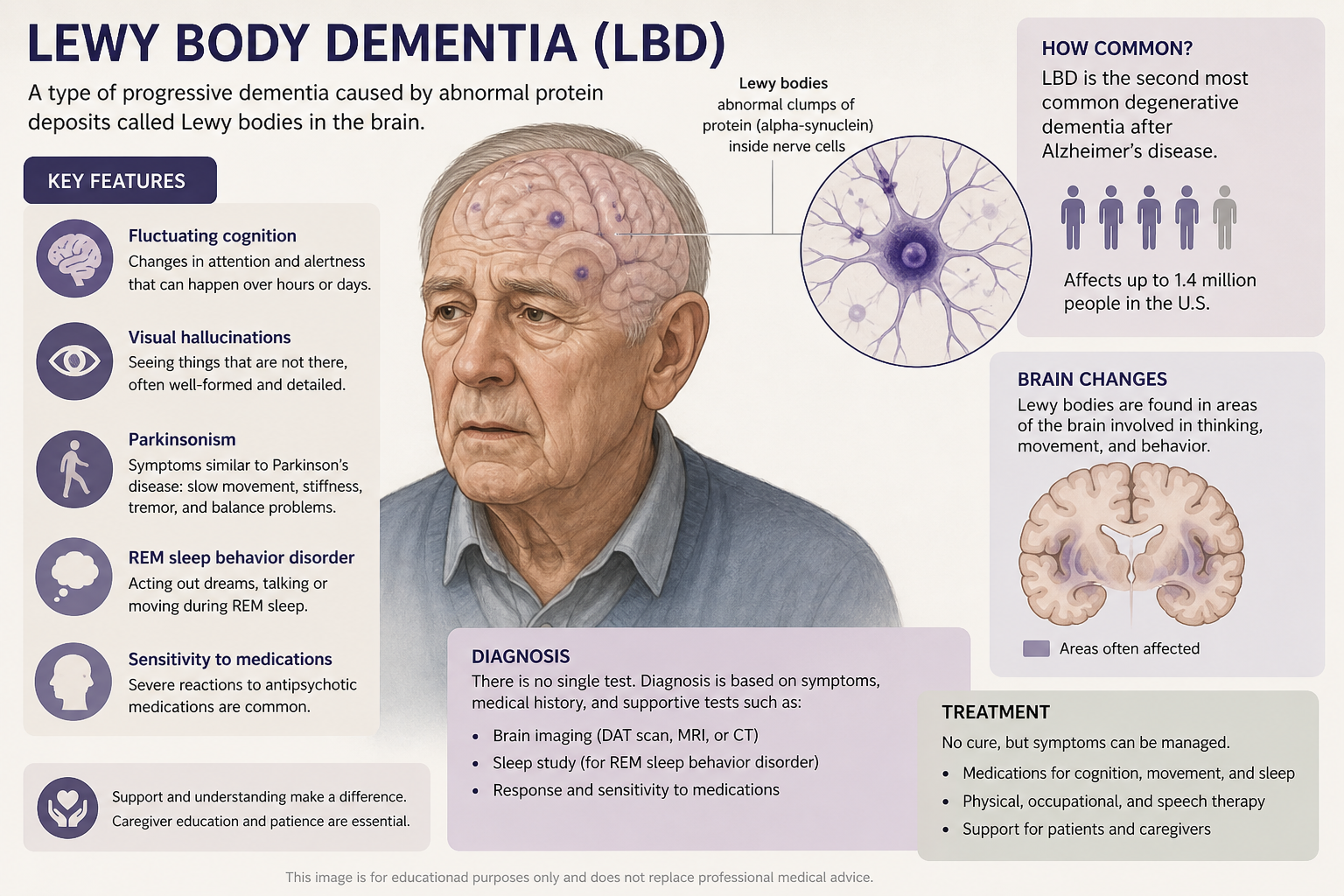
- 95% sensitivity for Lewy body dementia: 19 of 20 LBD patients tested positive for alpha-synuclein seeding activity in CSF — high enough to support real diagnostic use.
- 93.5% specificity, 99.4% negative predictive value: A negative result was strong evidence against Lewy body disease in this clinical setting; positive results still need context.
- 15.8% of Alzheimer’s cases hid Lewy body biology: 32 of 203 clinically diagnosed Alzheimer’s patients were alpha-synuclein-positive — co-pathology routine biomarkers missed.
- Standard CSF and blood markers explained little: Within the Alzheimer’s group, only the CSF Aβ 42/40 ratio differed between alpha-synuclein-positive and -negative patients. GFAP, NfL, and tau did not.
- Frontotemporal dementia was clean: Zero of 30 FTD patients tested positive — evidence the assay is not a generic neurodegeneration alarm.
- 398 routine memory-clinic patients: ALZAN cohort, Montpellier–Nimes–Perpignan, November 2022 to July 2024. Tested in the diagnostic gray zone clinics actually face.
Source: medRxiv (2026) | Jourdan et al.
Dementia diagnosis is often presented as a sorting exercise — Alzheimer’s here, Lewy body dementia there, frontotemporal dementia somewhere else. Biology refuses to cooperate. Many brains carry more than one disease process at the same time, and clinical labels keep treating mixed pathology like a rounding error.
This study, run inside ordinary French memory clinics, tested whether a CSF assay for alpha-synuclein seeding activity could surface that hidden biology in real patients — not autopsy-confirmed cases. It worked clearly enough in Lewy body dementia, and uncomfortably well in Alzheimer’s.
The Protein That Does Not Stay in Its Lane
Alzheimer’s disease is biologically defined by amyloid and tau. Lewy body dementia is defined by alpha-synuclein, the misfolded protein that forms Lewy bodies and Lewy neurites. In real patients, those borders blur constantly. Autopsy series have shown for years that alpha-synuclein appears in Alzheimer’s brains and amyloid-tau pathology appears in Lewy body brains. The clinical problem is that autopsy answers come too late to guide care.
Seed amplification assays work around that timing problem. They take cerebrospinal fluid, expose it to a controlled lab reaction, and ask whether anything in the sample can trigger normal alpha-synuclein to misfold and aggregate. If it can, the sample is positive — meaning Lewy-type biology is active in that brain right now.
The principle is borrowed from prion biology. A tiny amount of misfolded protein acts as a seed, recruiting normal protein into the same shape. The assay amplifies that process enough to detect in a fluorescence readout. RT-QuIC — real-time quaking-induced conversion — runs the reaction in quadruplicate and applies predefined positivity thresholds.
A Cohort That Looked Like the Real Diagnostic Mess
The ALZAN cohort enrolled 398 patients from routine memory clinics in Montpellier, Nimes, and Perpignan between November 2022 and July 2024. Each had CSF and blood collected, MMSE testing, and clinical diagnoses confirmed independently by two senior neurologists.
The mix is what makes the study useful. It was not a tidy autopsy-confirmed case series; it was the diagnostic gray zone:
- Alzheimer’s disease: 203 patients
- Lewy body dementia: 20 patients
- Frontotemporal dementia: 30 patients
- “Other” diagnoses and mixed presentations: 138 patients
This is the population the assay would actually serve in clinical practice — heterogeneous, partly uncertain, clinically labeled but biologically ambiguous.
The Lewy Body Signal Was Hard to Miss
The headline result was clean. Nineteen of 20 Lewy body dementia patients tested positive — 95% sensitivity, with 93.5% specificity against patients who carried neither LBD nor Alzheimer’s disease. The negative predictive value reached 99.4%, which means a negative result was strong evidence against Lewy body biology in this setting.
The frontotemporal dementia group is the other clean signal worth noting. Zero of 30 FTD patients tested positive. That does not guarantee perfect specificity in every population, but it argues the assay is detecting Lewy-type biology specifically rather than acting as a generic neurodegeneration alarm.
The Alzheimer’s Result Is the Clinically Provocative One
32 of 203 patients carrying an Alzheimer’s diagnosis tested positive for alpha-synuclein seeding activity — 15.8%. These are not patients in whom the Alzheimer’s diagnosis was wrong. They are patients in whom it was incomplete.
That number lines up with what autopsy series have been showing for years, but until now there was no live-patient way to surface it. Hidden Lewy-body biology helps explain a familiar clinical puzzle: why patients with apparently identical Alzheimer’s biomarker profiles can progress differently, develop hallucinations, show parkinsonian features, or have REM-sleep behavior disorder that does not fit a clean Alzheimer’s template.
The label “Alzheimer’s disease” may still be right for those patients. It just stops being the whole story.
Standard Biomarkers Were Not Enough to See This
The cohort-wide comparison showed expected differences — alpha-synuclein-positive patients had lower MMSE, lower CSF Aβ 42/40 ratios, and higher plasma GFAP — but those gaps largely reflected the diagnostic mix across positive and negative groups.
The narrower comparison is the one that matters: within the Alzheimer’s group, alpha-synuclein-positive and -negative patients differed only in CSF Aβ 42/40, a marker of greater amyloid burden. GFAP, neurofilament light, total tau, and phosphorylated tau did not separate them.
That gap is the clinical point of the assay. Amyloid, tau, GFAP, and NfL are powerful markers, but none of them directly measure alpha-synuclein seeding. A patient can look like archetypal Alzheimer’s on the standard panel and still carry additional Lewy-body biology that none of those markers can see.
The “other” diagnostic group showed a 7.9% positivity rate — harder to interpret, since the category mixes neurological conditions, mood-driven cognitive complaints, and edge cases. The result reinforces a different point: this kind of test should be read alongside clinical context, not as a standalone verdict.
What This Could Change About How Dementia Is Diagnosed
The study did not prove that hidden alpha-synuclein independently worsens cognition in Alzheimer’s disease. The authors are direct about that limit. The cohort lacked longitudinal follow-up, and clinical features that typically accompany Lewy biology — hallucinations, fluctuations, REM sleep behavior disorder, sensitivity to antipsychotics — were not deeply analyzed.
What the result does support is a more layered approach. The right diagnostic question is moving from “which single label fits this patient best” to “which protein processes are active in this patient at the same time.” That shift has implications well beyond the clinic. Trials enrolling people on amyloid or tau status alone may be quietly absorbing alpha-synuclein co-pathology as noise — and explaining away genuine biological subgroups as treatment-response variability.
This is a preprint, so peer review will refine it. But the trajectory it implies is hard to argue with: dementia diagnosis as a biological fingerprint rather than a single sticker. Amyloid, tau, neurodegeneration markers, and alpha-synuclein seeding considered together — instead of forcing every patient into one clean label.
Citation: Jourdan et al. Integrating alpha-synuclein seeding activity into routine practice: insights from the multicenter ALZAN cohort. medRxiv. 2026. DOI: 10.64898/2026.04.21.26351389
Study Design: Multicenter prospective memory-clinic cohort study (preprint).
Sample Size: 398 patients — 203 Alzheimer’s disease, 20 Lewy body dementia, 30 frontotemporal dementia, 138 other diagnoses.
Key Statistic: 95% sensitivity, 93.5% specificity, 99.4% NPV for Lewy body dementia. 15.8% of Alzheimer’s-diagnosed patients were alpha-synuclein seed positive.