TL;DR: A 2026 study in Nature Neuroscience from Baylor College of Medicine showed that boosting Sox9, a transcription factor that regulates aging astrocytes, improved astrocytes’ ability to clear amyloid plaques in Alzheimer’s mouse models and preserved cognitive function — pointing to a treatment strategy that targets the brain’s own support cells rather than directly attacking amyloid.
Key Findings
- Sox9 overexpression improved astrocyte plaque clearance: Increasing Sox9 levels in astrocytes of Alzheimer’s mouse models significantly improved phagocytosis of amyloid-beta plaques.
- Cognitive function was preserved: Mice with boosted astrocyte Sox9 retained better cognitive function than untreated Alzheimer’s-model controls.
- Sox9 controls many aging-astrocyte genes: The transcription factor regulates a large set of genes that change with brain aging, making it a single intervention point that touches multiple downstream pathways at once.
- Astrocyte-targeted strategy, not amyloid-targeted: The approach boosts the brain’s own support cells rather than introducing antibodies that bind amyloid directly — a different therapeutic logic from the lecanemab/donanemab class.
- Astrocytes do more than support neurons: They participate in brain communication, memory storage, and clearance of damaged proteins. Their functional decline with aging may be a contributor to neurodegeneration that current Alzheimer’s drugs do not address.
- Mouse-model only, human translation untested: The findings come from genetically engineered Alzheimer’s mouse models. Whether the same Sox9 manipulation produces clinical benefit in humans is the next research step.
Source: Nature Neuroscience (2026) | Choi, Deneen et al.
Astrocytes are non-neuronal cells in the brain that support neurons through metabolic supply, neurotransmitter clearance, and direct participation in synaptic communication.
Their function changes substantially with brain aging, but the role of those changes in driving neurodegenerative disease has been understudied compared with neuron-focused mechanisms.
This study tested whether boosting one transcription factor that controls aging-astrocyte gene expression could push astrocytes back toward a younger functional state — and whether that shift would slow Alzheimer’s pathology in mice.
Sox9 as the Master Regulator of Aging-Astrocyte Gene Expression
The mechanistic premise of the study turns on the role of Sox9, a transcription factor:
- Sox9 controls many aging-astrocyte genes: A single transcription factor that influences expression of multiple downstream genes during astrocyte aging.
- Aging astrocytes show profound functional alterations: Including changes in plaque clearance, neurotransmitter handling, and inflammatory state.
- Single-target leverage: Manipulating one transcription factor can shift many downstream pathways at once, in contrast to interventions targeting individual proteins or pathways downstream.
First author Dr. Dong-Joo Choi described the framing:
“Astrocytes perform diverse tasks that are essential for normal brain function, including facilitating brain communications and memory storage. As the brain ages, astrocytes show profound functional alterations; however, the role these alterations play in aging and neurodegeneration is not yet understood.”
Boosting Sox9 in Mouse Astrocytes Improved Plaque Clearance
The team manipulated Sox9 expression specifically in astrocytes within Alzheimer’s mouse models.
The experimental design:
- Mouse models: Genetically engineered Alzheimer’s mouse models that develop amyloid plaques and cognitive deficits.
- Intervention: Sox9 expression boosted specifically in astrocytes.
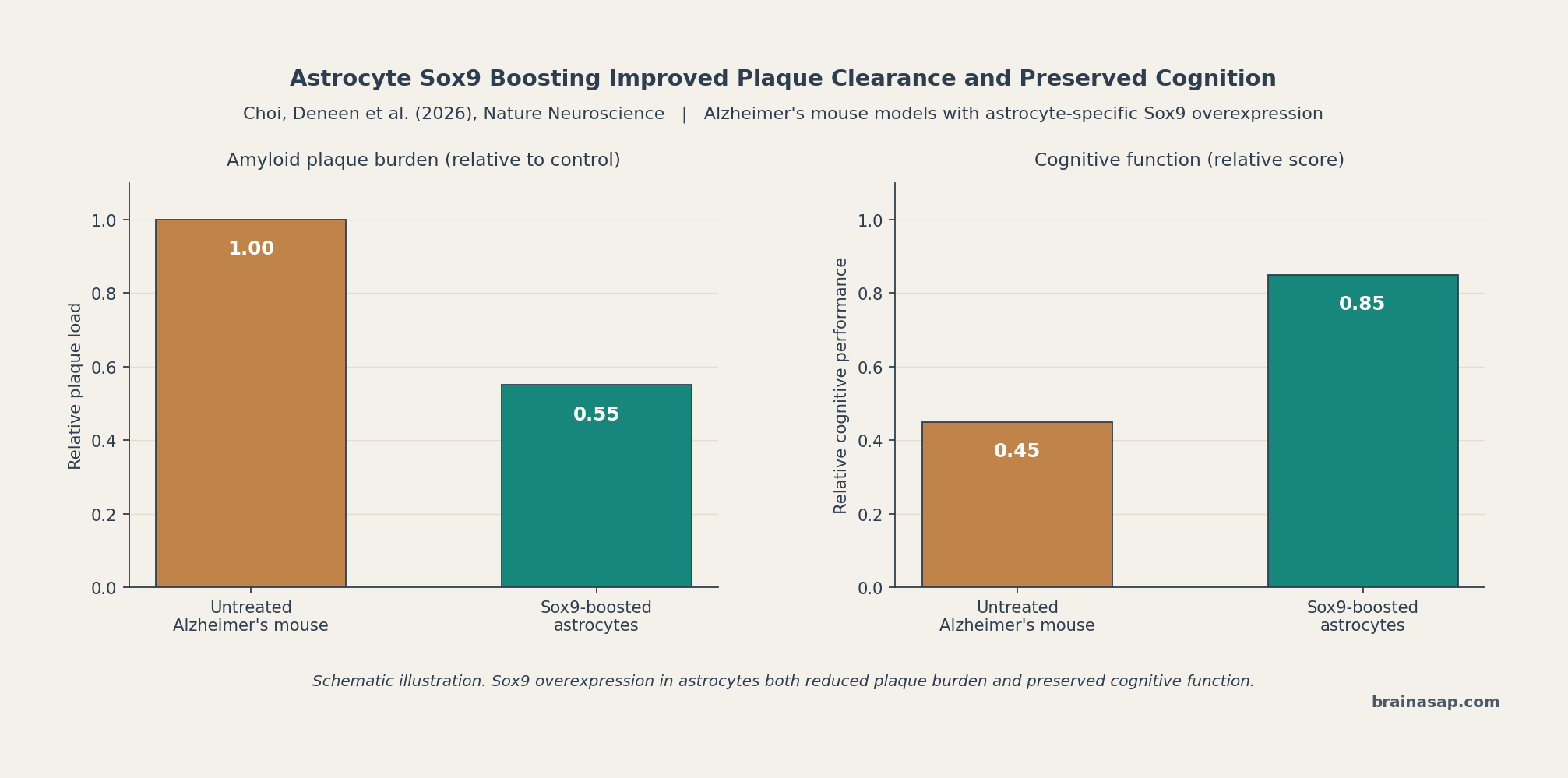
- Plaque outcome: Astrocytes with elevated Sox9 showed significantly improved phagocytosis of amyloid-beta plaques.
- Cognitive outcome: Mice with boosted astrocytic Sox9 retained better cognitive function compared with untreated Alzheimer’s-model controls.
The combination of mechanistic clearance evidence (plaques removed) and behavioral evidence (cognition preserved) is what gives the result its weight. A treatment that cleared plaques without producing functional benefit would just be repeating the disappointing pattern of monoclonal antibody trials.
Why an Astrocyte-Targeted Strategy Is Different From Anti-Amyloid Antibodies
The dominant Alzheimer’s pharmaceutical strategy has been monoclonal antibodies (lecanemab, donanemab) that bind amyloid directly and help clear it. The recent Cochrane review concluded the antibody class produced no clinically meaningful effect in mild Alzheimer’s.
The Sox9 strategy is mechanistically different:
- Targets the brain’s own support cells: Boosts astrocyte function rather than introducing exogenous antibodies.
- Restores function rather than substituting for it: Pushes astrocytes back toward a younger, more efficient phagocytic state.
- Single transcription factor with broad downstream effects: Targeting Sox9 may produce coordinated changes across multiple aging-related astrocyte pathways simultaneously.
- Addresses an aspect of disease the antibody approach does not: Astrocyte dysfunction with aging is a separate contributor to neurodegeneration that pharmaceutical efforts have largely ignored.
That difference in approach is the reason the work is positioned as a candidate for further translational investment, not as another iteration of the antibody strategy.
Mouse-Model Evidence Only, Translation Is the Open Question
- Mouse-to-human translation is not automatic: Many candidates that improved Alzheimer’s pathology in mouse models have failed to produce clinical benefit in human trials. The species gap is the largest unproven step.
- Sox9 manipulation in human astrocytes is technically harder: Genetic boosting of one transcription factor in mice via standard tools is straightforward; doing the equivalent in human patients requires gene therapy or pharmacological mimics that have not been developed for this target.
- Brain-wide vs region-specific effects unclear: The astrocyte intervention in mice was applied broadly. Whether broader Sox9 modulation in humans would produce off-target effects in non-disease brain regions is unknown.
- Aging-astrocyte heterogeneity not fully characterized: Astrocytes are functionally diverse; whether all astrocyte subtypes respond similarly to Sox9 boosting is an open question.
- Cognitive measures in mice are limited: Mouse cognitive tests probe a narrow slice of what human Alzheimer’s disrupts. Functional preservation in mice does not automatically map to memory, language, and daily-living preservation in humans.
Astrocyte-Targeted Therapy Joins the Alzheimer’s Pipeline as a Distinct Mechanistic Class
The implications follow from the mechanism-distinct framing:
- Distinct mechanistic class worth pursuing: Given the limited clinical results of anti-amyloid antibodies, alternative mechanisms with mouse-model evidence and cognitive preservation deserve investment.
- Sox9 modulation is the lever, not necessarily a drug yet: Translation requires either gene-therapy delivery or small-molecule mimics that boost Sox9-pathway activity in human astrocytes.
- Combination strategies are plausible: Astrocyte boosting and amyloid clearance are mechanistically independent; pairing the two approaches in future trials is a natural design.
- Reframes the field’s view of astrocytes in Alzheimer’s: The work argues that aging-driven astrocyte dysfunction is not just a downstream consequence of disease but a contributor that can be intervened on directly.
Citation: DOI: 10.1038/s41593-025-02115-w. Choi DJ, Deneen B et al. Astrocytic Sox9 overexpression in Alzheimer’s disease mouse models promotes Aβ plaque phagocytosis and preserves cognitive function. Nature Neuroscience. 2026.
Study Design: Genetic manipulation of Sox9 expression in astrocytes within Alzheimer’s mouse models; outcome assessment included amyloid-beta plaque burden and cognitive function.
Sample/Model: Genetically engineered Alzheimer’s mouse models with astrocyte-specific Sox9 overexpression.
Key Statistic: Sox9 overexpression in astrocytes significantly improved amyloid plaque phagocytosis and preserved cognitive function compared with untreated Alzheimer’s-model controls.
Caveat: Mouse-model evidence only; mouse-to-human translation is the largest unproven step; Sox9 manipulation in humans requires gene therapy or pharmacological mimics not yet developed; off-target effects on non-disease astrocyte populations untested.