TL;DR: A 2026 medRxiv preprint tested SimulAD, an Alzheimer’s disease progression simulator, and reported that it reproduced mean amyloid-clearance results within 5% error across six major phase III anti-amyloid trials while also separating positive trials from null trials in retrospective power analyses.
Key Findings
- Six phase III trials: Researchers retrospectively simulated TRAILBLAZER-ALZ2, CLARITY AD, EMERGE, ENGAGE, GRADUATE I, and GRADUATE II with one trained disease-progression model.
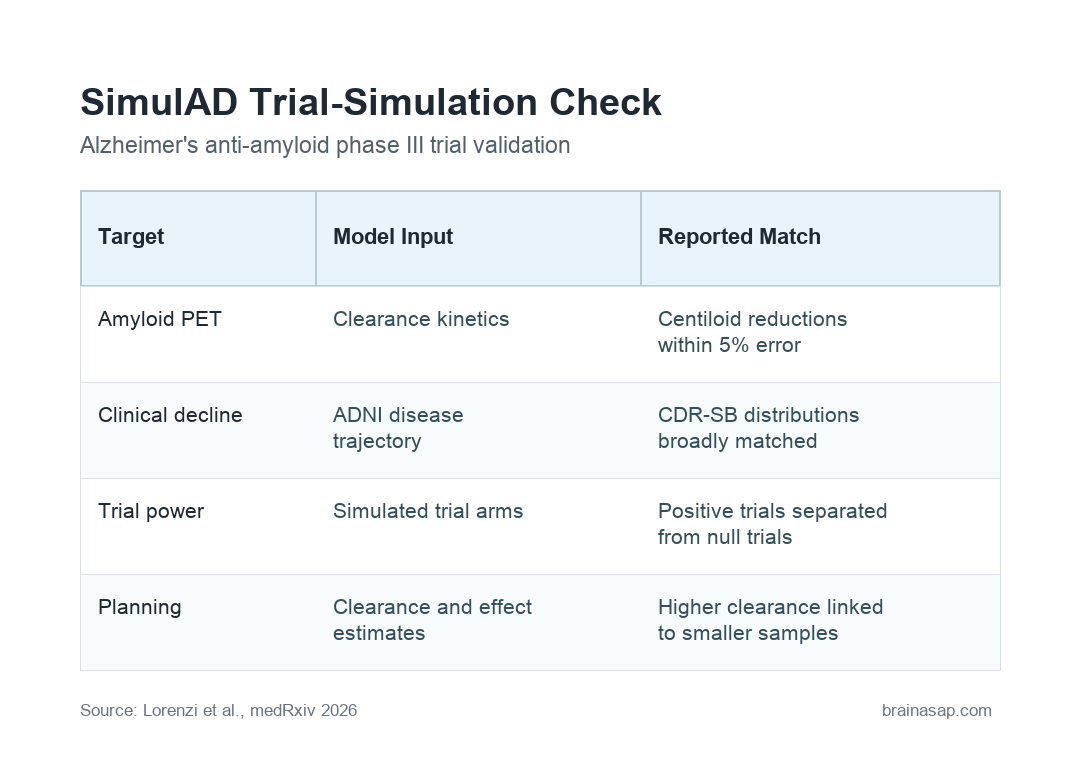
- Within 5% amyloid-clearance error: SimulAD reproduced published mean centiloid reductions within 5% error across all six trial simulations.
- CDR-SB was used for clinical decline: The model generated Clinical Dementia Rating-Sum of Boxes (CDR-SB), a scale of dementia-related cognitive and daily-function impairment, distributions broadly consistent with reported placebo and treatment-arm outcomes.
- Positive and null trials separated: In retrospective power analysis, calibrated simulations separated the three positive trials from the three null trials, with EMERGE near the decision boundary.
- Clearance kinetics mattered: Higher amyloid-clearance rates were associated with larger calibrated clinical effects and lower estimated sample sizes.
Source: medRxiv (2026) | Lorenzi et al.
SimulAD is a computer model for Alzheimer’s disease progression, not a new drug. It simulates whether a specific amyloid-clearance pattern is likely to be followed by changes in tau, structural MRI measures, and cognition.
Recent antibody trials have made this trial-design question more urgent: how much amyloid clearance, how fast, and in how many patients might be needed before a phase III study can detect a meaningful slowing of cognitive decline?
SimulAD Modeled Alzheimer’s Disease Progression Across Biomarkers
Most trial-simulation tools in drug development start from pharmacokinetics and pharmacodynamics: how much drug reaches the body, how the body handles it, and how a target biomarker changes.
For anti-amyloid trials, a PK/PD-only framework is too narrow by itself because the clinical question is not only whether amyloid PET changes; it is whether amyloid clearance is followed by meaningful shifts in tau biology, neurodegeneration, brain structure, and cognition.
SimulAD was built around the gap between amyloid removal and clinical trajectory: a treatment can clear amyloid while later outcomes still depend on the timing and size of downstream biomarker changes.
Researchers trained SimulAD on longitudinal observational data from the Alzheimer’s Disease Neuroimaging Initiative (ADNI). ADNI is a long-running research dataset with repeated biomarker, imaging, and cognitive measurements.
ADNI’s repeated measurements make it suitable for modeling disease trajectories over time.
Researchers emphasized that the model was trained without access to trial-arm labels or drug-specific outcomes. The no-trial-label training is central to the claim: the simulator was not fit directly to the anti-amyloid trial results it later tried to reproduce.
- Amyloid: The model encoded intervention as piecewise amyloid-clearance terms, allowing simulated treatment to remove amyloid at different rates.
- Tau and MRI: It jointly modeled downstream tau and structural MRI trajectories rather than treating amyloid as a standalone endpoint.
- Cognition: It also modeled cognitive trajectories, including CDR-SB outcomes used in Alzheimer’s clinical trials.
Six Anti-Amyloid Trials Tested the Simulator Retrospectively
The strongest part of the preprint is the retrospective validation design. Researchers did not test SimulAD on one convenient trial.
They simulated six landmark phase III anti-amyloid programs that included both successful and unsuccessful outcomes.
The trial set included TRAILBLAZER-ALZ2, CLARITY AD, EMERGE, ENGAGE, GRADUATE I, and GRADUATE II. These trials differ in antibody program, clearance kinetics, clinical results, and statistical interpretation, which gives the simulator a more demanding target than a single case study.
For each trial, calibration was limited to amyloid-clearance kinetics. In practical terms, the simulator was told how amyloid was cleared in that trial context, then it had to generate plausible downstream biomarker and clinical trajectories from its disease-progression structure.
- TRAILBLAZER-ALZ2 and CLARITY AD: These trials served as positive examples in the retrospective power analysis.
- GRADUATE I and GRADUATE II: These trials served as null examples that the simulator needed to keep below the positive-trial range.
- EMERGE and ENGAGE: These aducanumab trials were especially informative because the reported trial program has been difficult to interpret, and the simulation placed EMERGE near the decision boundary while ENGAGE stayed below it.
Amyloid Centiloid Reductions Matched Published Trial Results
The cleanest quantitative result was amyloid clearance. SimulAD reproduced published mean centiloid reductions within 5% error across all six simulated trials.
Centiloids are a standardized amyloid positron emission tomography (PET) scale. A lower centiloid value means less amyloid signal on PET imaging.
Researchers can use that scale to compare amyloid-lowering treatment effects across trials and drugs.
This is an important first gate for the model. If a simulator cannot reproduce the biomarker it is directly calibrated around, it has little chance of giving credible downstream clinical estimates.
Here, amyloid clearance was not the whole claim, but it was the anchor.
Clinical decline was the harder test. Researchers reported that SimulAD generated placebo and treated-arm CDR-SB distributions that were broadly consistent with the published trial outcomes.
CDR-SB stands for Clinical Dementia Rating-Sum of Boxes. It combines ratings across memory, orientation, judgment, community affairs, home and hobbies, and personal care.
Higher scores mean more impairment, so slower CDR-SB worsening is the kind of clinical effect anti-amyloid trials are trying to show.
Retrospective Power Analysis Separated Positive From Null Trials
A trial simulator has more value when it can help with design decisions, not just recreate biomarker curves. Researchers therefore ran retrospective power analyses.
Those analyses asked whether simulated trial outcomes would have been statistically likely to detect a treatment effect.
Those simulations separated the three positive trials from the three null trials. TRAILBLAZER-ALZ2 and CLARITY AD landed on the positive side, while ENGAGE and both GRADUATE trials stayed below it.
EMERGE sat near the decision boundary, which fits its unusual position in the anti-amyloid evidence base.
For trial planning, the boundary behavior is important. The model did not simply label every amyloid-lowering trial as clinically positive.
Instead, amyloid clearance rate, downstream disease trajectory, and sample-size assumptions had to line up.
- Higher clearance: Faster or more complete amyloid reduction was associated with larger calibrated clinical effects.
- Lower sample-size estimates: Stronger simulated effects reduced the estimated number of participants needed to detect a difference.
- Borderline programs: EMERGE near the boundary shows why simulation should be treated as decision support, not a verdict machine.
Why a Disease-Cascade Simulator Could Help Alzheimer’s Trials
Alzheimer’s treatment development is expensive because phase III trials need large samples, long follow-up, complex imaging, and clinically meaningful endpoints.
A disease-cascade simulator could help researchers test design assumptions before committing to a full trial.
The key advantage is multi-modal modeling. SimulAD does not treat amyloid as a separate biomarker detached from the rest of the disease.
It links amyloid, tau, structural MRI, and cognition in one latent ordinary differential equation system.
SimulAD’s linked amyloid-tau-MRI-cognition model could support several planning decisions. A sponsor could ask whether a faster amyloid-clearance schedule should change sample size, whether a trial needs longer follow-up, or whether a biomarker response is large enough to plausibly translate into a CDR-SB difference.
Practical interpretation: The preprint supports SimulAD as a quantitative planning tool for anti-amyloid trials, especially when trial design depends on clearance kinetics and expected clinical power.
The Main Caveat Is That This Is a Preprint Simulator Study
This source should not be read as proof that any particular anti-amyloid drug works better than another. The study is a simulation and validation exercise, posted as a preprint.
The available captured source material provides abstract-level detail rather than a full independent methods audit.
Several limits follow from the preprint’s simulation design. SimulAD depends on ADNI-trained observational trajectories, assumptions about the amyloid cascade, and calibration to trial-specific amyloid-clearance kinetics.
Those choices may perform differently in future trials, different patient populations, or therapies that act outside amyloid clearance.
- Preprint status: The work had not completed journal peer review at the time of posting.
- Model dependence: The conclusions depend on how well ADNI-trained trajectories represent trial populations.
- Amyloid-centered scope: The simulator is most directly suited to anti-amyloid interventions and may not generalize to treatments with different mechanisms.
- Clinical-use limit: This is a research-planning tool, not a patient-level prediction model for clinical care.
The finding is still relevant because it addresses a real bottleneck. Alzheimer’s trials need better ways to connect biomarker movement with clinical power.
Retrospective validation across positive and null phase III trials is a stronger starting point than a simulator shown only on synthetic examples.
The next test is prospective. If SimulAD can predict trial behavior before results are known, its value would become much clearer.
Citation: DOI: 10.64898/2026.05.03.26352317; Lorenzi et al.; A data-driven Alzheimer’s disease progression simulator for retrospective validation and prospective Phase III power design; medRxiv; 2026;2026.05.03.26352317.
Study Design: Preprint simulation study using an ADNI-trained Alzheimer’s disease progression model and retrospective validation against six phase III anti-amyloid trials.
Sample/Model: Longitudinal ADNI observational data for model training, with retrospective simulations of TRAILBLAZER-ALZ2, CLARITY AD, EMERGE, ENGAGE, GRADUATE I, and GRADUATE II.
Key Statistic: SimulAD reproduced published mean amyloid centiloid reductions within 5% error across all six simulated trials.
Caveat: Preprint simulator validation; prospective trial prediction and full peer-reviewed methods review are still needed.