TL;DR: A 2026 World Journal of Biological Psychiatry study found brain-wave connectivity between a prefrontal control area and a depression-linked cingulate region differed in patients who later remitted after rTMS magnetic stimulation.
Key Findings
- Remitters had lower pre-treatment DLPFC-to-sgACC alpha connectivity: Baseline alpha-band isolated effective coherence (iCoh) — a directional measure — from left DLPFC to subgenual anterior cingulate cortex was significantly lower in patients who subsequently remitted.
- The finding was directional, not just correlational: iCoh captures directed information flow from one region to another — a more mechanistically specific claim than generic synchrony between DLPFC and sgACC.
- The two regions are the core treatment circuit: Left DLPFC is the rTMS stimulation target; sgACC is one of the key mood-circuit nodes linked to depressive-state persistence and antidepressant response. The biomarker tracks the actual therapeutic pathway.
- Lower connectivity may mark a circuit with room to be shifted: One interpretation is that lower pre-treatment coupling findings a less-locked-in circuit state that rTMS can reorganize more effectively.
- Still a candidate-stage finding: 30-patient observational cohort, single center — needs prospective replication with a pre-registered pipeline before it can guide treatment selection.
Source: World Journal of Biological Psychiatry (2026) | Shimizu et al.
Repetitive transcranial magnetic stimulation for depression has a prediction problem.
rTMS works for a meaningful fraction of patients — but treatment takes weeks of repeated clinic visits, and there’s no reliable way beforehand to know whether a given patient will respond.
That uncertainty is costly: it keeps people in a failing treatment longer than necessary and delays access to alternatives.
The field has been looking for a pre-treatment biomarker that could stratify patients before the clock starts.
This small but mechanistically targeted study found one candidate: a directed connectivity measure between the cortical stimulation target and the deeper mood circuit it’s trying to reach.
DLPFC-to-sgACC Direction Made EEG Connectivity Predictive for rTMS
Most EEG biomarker studies in depression end with a connectivity difference between responders and non-responders — usually a region-pair that shows more or less synchrony. Those findings can advance the science without being clinically actionable, because correlated activity between two regions doesn’t tell you what to do about it.
This study used isolated effective coherence (iCoh), which estimates directed information flow while minimizing indirect paths that can confuse interpretation. The claim is more specific: before rTMS begins, left DLPFC was sending lower alpha-band findings toward the subgenual anterior cingulate cortex (sgACC) in patients who would later remit.
The directionality matters because these aren’t arbitrary regions — they’re the two nodes of the actual rTMS treatment circuit. The DLPFC is where the coil is placed.
The sgACC is the deeper mood node that rTMS is trying to modulate by changing the network above it. A pre-treatment measure of DLPFC-to-sgACC connectivity is close to the mechanism in a way that a whole-brain connectivity map is not.
Lower Coupling Before Treatment May Predict rTMS Response
The finding that remitters had lower directed DLPFC-to-sgACC alpha coherence before treatment raises a mechanistic possibility. High-frequency rTMS is meant to excite the left DLPFC, which then propagates through the network to modulate downstream mood circuitry.
If that pathway is already locked into a strong, stable pattern before treatment, rTMS may have less capacity to shift it.
Lower pre-treatment coupling could mark a circuit state that’s more plastic — less entrenched, more responsive to stimulation-driven reorganization.
That’s speculative from 30 patients, but it’s a testable prediction that’s mechanistically coherent with what rTMS is supposed to accomplish.
Alpha-band activity is also worth noting specifically. Alpha rhythms are often associated with inhibition, gating, and cortical state.
Directed alpha flow from DLPFC to sgACC could reflect how strongly the prefrontal control network is coupling to — or inhibiting — the affect-regulation circuitry below it.
Whether that reflects a depressive circuit state or an individual brain architecture trait (or both) is something larger studies with clinical subtype breakdowns could begin to answer.
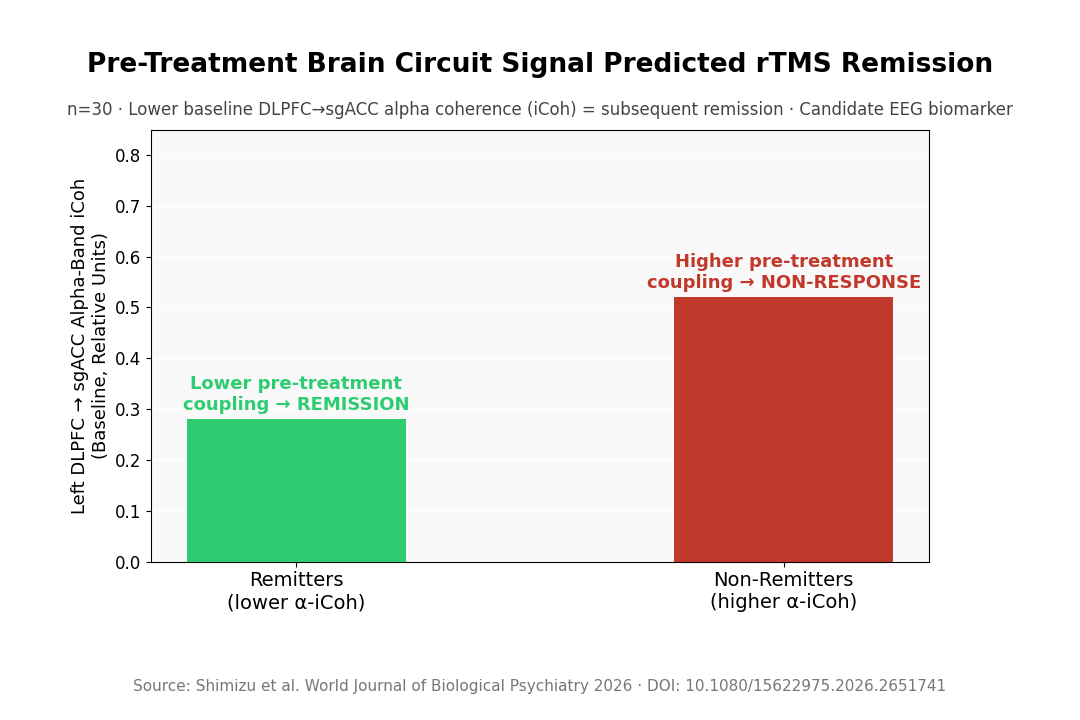
Nonremitters Showed Stronger Baseline DLPFC-to-sgACC Coupling
Prediction studies are usually framed around identifying responders. But for rTMS — which takes weeks, requires repeat visits, and carries emotional stakes for people who’ve already failed medications — the nonresponse side may matter just as much.
A pre-treatment biomarker that flags lower probability of remission with standard left-DLPFC rTMS isn’t a dead end for the patient. It could be the prompt to consider a different stimulation target (the right DLPFC, bilateral protocols, or deeper-circuit approaches), a different stimulation frequency, or a different treatment pathway entirely.
Early identification of likely non-responders accelerates getting to the right treatment rather than wasting weeks on the wrong one.
rTMS EEG Connectivity Needs Larger Prospective Replication
Thirty patients, one center, retrospective analysis: this is a candidate biomarker, not a clinical tool.
The next study needs to be prospective — measure iCoh before outcomes are known, commit to a pre-registered EEG processing pipeline, define remission criteria before unblinding, and test in an independent cohort.
The practical challenge is that rTMS protocols vary considerably. Coil placement, stimulation frequency, motor-threshold methods, treatment duration, and medication background can all affect outcomes.
A biomarker that works only within one center’s specific setup will be scientifically plausible and clinically fragile. The replication should vary some of these parameters deliberately to test robustness.
If the iCoh finding survives those challenges, EEG-guided rTMS selection becomes a concrete clinical possibility — a 30-minute brain recording before starting treatment, rather than weeks of treatment before knowing whether it’s working. That would change the calculus of rTMS meaningfully for both patients and services.
Citation: DOI: 10.1080/15622975.2026.2651741. Shimizu et al. Reduced pre-treatment isolated effective coherence between the dorsolateral prefrontal cortex and the subgenual anterior cingulate cortex as a potential predictive marker for remission following repetitive transcranial magnetic stimulation in major depressive disorder. World Journal of Biological Psychiatry. 2026.
Study Design: Single-center observational rTMS biomarker study; pre-treatment EEG source modeling, left DLPFC high-frequency rTMS protocol.
Sample Size: 30 adults with major depressive disorder.
Key Statistic: Lower baseline alpha-band iCoh from left DLPFC to sgACC was associated with subsequent rTMS remission. Result needs prospective validation before clinical use.