TL;DR: A 2026 medRxiv preprint found that ex-smokers had higher fMRI blood oxygen level dependent (BOLD) responses to high-energy food pictures than adults with obesity or abstinent alcohol dependence, suggesting a brain reward route for post-quitting weight gain.
Key Findings
- 77-person fMRI comparison: Researchers compared 25 ex-smokers, 26 abstinent adults with alcohol dependence, and 26 adults with obesity who were actively dieting.
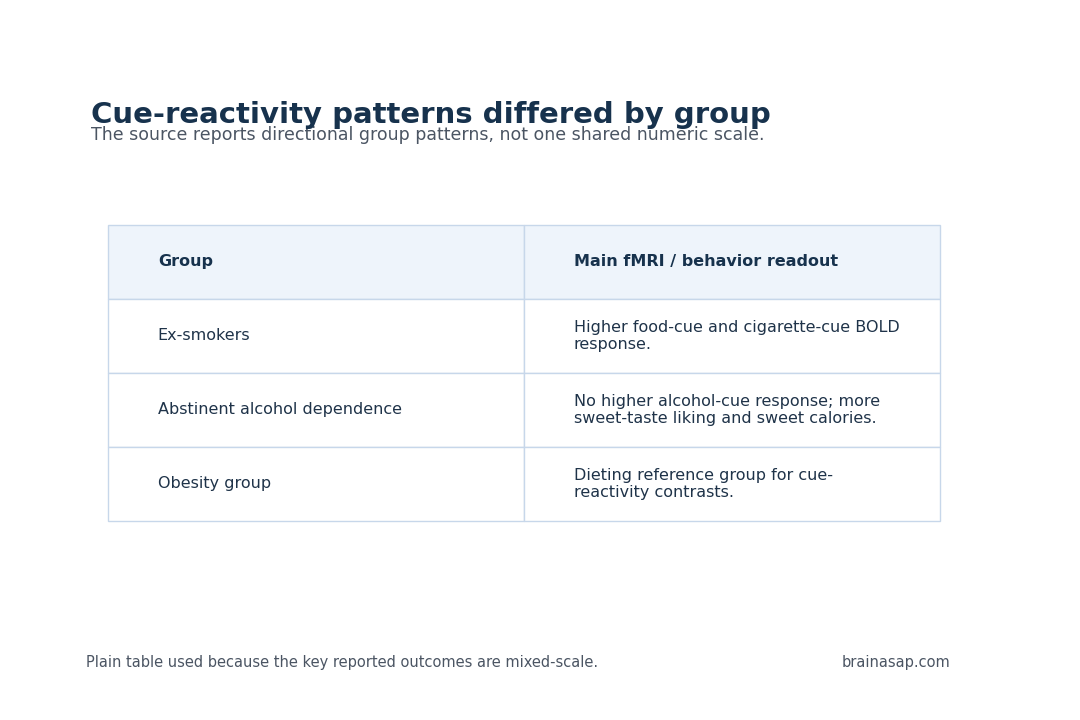
- Food cues activated reward regions in ex-smokers: Ex-smokers had greater BOLD response to high-energy food pictures than the obesity and alcohol-abstinence groups.
- Cigarette cues persisted without higher appeal ratings: Ex-smokers also showed greater response to cigarette pictures in frontal and insula regions compared with the obesity group.
- Alcohol cue differences were absent: The study found no group differences in preferred alcohol cue reactivity.
- Sweet preference appeared in alcohol abstinence: The abstinent alcohol-dependence group rated sweet taste as more pleasant and consumed more sweet-dish calories.
Source: medRxiv (2026) | GHADD study team
Food Cue fMRI Responses Were Higher in Ex-Smokers
Weight gain after smoking cessation is often described through appetite, metabolism, or willpower. This neuroimaging study examined a more specific possibility: the brain’s reward response to high-energy food cues can remain heightened after quitting cigarettes, even when people are no longer actively smoking.
Researchers used the Gut Hormones in ADDiction (GHADD) neuroimaging study to compare three clinical populations. The groups were 25 ex-smokers with prior nicotine use disorder, 26 abstinent adults with alcohol dependence, and 26 adults with obesity who were actively dieting. Participants completed an fMRI cue-reactivity task using high-energy food, preferred alcohol, and cigarette images.
The main contrast was not based on self-reported appeal alone. Ex-smokers showed higher BOLD responses to high-energy food pictures in reward-processing regions, even without a matching difference in appeal ratings. BOLD is an fMRI signal related to changes in blood oxygenation, used as an indirect readout of neural activity.
Ex-Smokers Also Retained Cigarette Cue Responses
The ex-smoker group also responded more strongly to cigarette pictures than the obesity group in frontal gyrus, orbitofrontal cortex, frontal pole, and insula regions. These areas are relevant to valuation, salience, interoception, and cognitive control. The finding suggests that some drug-cue responsiveness can persist during medium-term abstinence even when craving is not obvious.
A person can report little craving in the scanner while the brain still allocates extra salience to smoking-related images. The same person can also show heightened response to high-energy food cues.
Together, those findings support a shared reward-salience mechanism that can complicate smoking cessation by making food cues more compelling.
- Food images: Ex-smokers had greater BOLD response to high-energy food pictures than both comparison groups.
- Cigarette images: Ex-smokers had greater response than the obesity group in frontal and insula regions.
- Subjective ratings: The neural differences appeared without matching appeal-rating differences.
Ex-smokers are not destined to gain weight. Post-cessation eating changes can still have a measurable brain-cue component, especially in environments full of high-energy food cues.
The absence of higher appeal ratings is part of the clinical value. A person can rate food images as no more appealing while fMRI still shows stronger activation in reward and salience circuitry. That gap helps explain why simple self-report can miss risk states during abstinence.
Alcohol Abstinence Showed a Sweet Taste Profile Instead
The alcohol-abstinence group showed a different behavioral profile. Researchers did not find group differences in preferred alcohol cue reactivity, but the abstinent alcohol-dependence group rated sweet taste as more pleasant and consumed more calories from sweet dishes during an ad libitum meal than the other groups.
That result fits a long-standing clinical observation: some people reduce alcohol use and increase preference for sweet foods. The study frames this as a possible sweet taste preference endophenotype, meaning a trait-like vulnerability that can relate to alcohol use disorder rather than simply to current drinking.
The meal test gives the alcohol finding behavioral weight. Participants were not only rating sweetness in the abstract; the abstinent alcohol-dependence group consumed more calories from sweet dishes during an ad libitum meal. The sweet-preference readout is therefore more clinically concrete than a questionnaire-only result.
The alcohol result also helps separate cue reactivity from eating behavior. The study did not find a stronger preferred-alcohol cue response, yet the same group showed a sweet-taste and sweet-intake profile. Recovery care should track both drug cues and substitute reward behaviors.
Those behaviors are clinically relevant because sweet foods can enter a recovery routine. They can reduce immediate alcohol craving for some people while still affecting weight, glucose control, sleep, and mood over time.
Smoking Cessation May Need Food-Cue Support
The treatment implication is not that food is another addiction in a simple one-to-one way. The data support a narrower point: ex-smokers can remain neurally reactive to salient cues, including high-energy food, after medium-term abstinence from cigarettes. That helps explain why weight gain after quitting is common and why advice based only on calorie counting can miss the reward-cue component.
Interventions can respond to that biology in several ways. Some patients benefit from planning for high-risk food contexts during the same period when they are managing smoking cues. Others need behavioral strategies that reduce cue exposure, change automatic eating routines, or pair cessation care with sleep and appetite support.
Medication and nutrition counseling also need to be coordinated rather than handled as separate issues after weight gain has already occurred.
For researchers, the next test is prediction. A stronger food-cue BOLD response should be followed prospectively to see whether it forecasts weight change, relapse vulnerability, or response to behavioral support. Without that follow-up, the scan result remains a mechanism candidate rather than a clinical decision tool.
The study also suggests a better outcome battery for cessation trials. Weight change alone is too blunt; future work should combine food-cue fMRI, actual meal intake, appetite ratings, craving reports, and abstinence duration. That would show whether the reward-cue signal explains behavior or simply travels alongside it.
That design would also help identify who needs extra support. Not every ex-smoker will have elevated food-cue response, and not every elevated response will translate into weight gain.
- Behavioral planning: Identify high-energy food cue environments before quit attempts or during abstinence maintenance.
- Relapse prevention: Treat food-cue reactivity and cigarette-cue reactivity as overlapping salience problems for some ex-smokers.
- Alcohol recovery care: Watch for sweet-taste preference and sweet-food intake in abstinent alcohol-use-disorder patients.
Small Group fMRI Needs Replication Before Clinical Use
The study has clear limits. Each group had 25 or 26 participants, so the findings need replication in larger cohorts. Group definitions also differed: ex-smokers, abstinent alcohol-dependence participants, and actively dieting adults with obesity are clinically distinct groups, not interchangeable models of reward biology.
fMRI cue-reactivity studies also require careful interpretation. BOLD responses are indirect and context-dependent. A stronger response to an image does not automatically predict relapse, calorie intake, or long-term weight change for a given person.
The study is better viewed as a mechanistic clue than as a patient-level prediction tool.
Still, the contrast is clear enough to shape future work: after smoking cessation, the brain can remain unusually responsive to high-energy food cues, while abstinent alcohol dependence can show a separate sweet-taste and sweet-intake profile. Those two readouts point toward different intervention needs, even though both sit under the wider umbrella of reward and salience.
Citation: DOI: 10.64898/2026.03.13.26348339; Salient cue reactivity and eating behaviours in ex-smokers, abstinent alcohol use disorder and obesity; medRxiv; 2026.
Study Design: Cross-sectional fMRI cue-reactivity and eating-behavior comparison across three clinical groups.
Sample Size: 25 ex-smokers, 26 abstinent adults with alcohol dependence, and 26 adults with obesity.
Key Result: Ex-smokers had greater BOLD response to high-energy food pictures than the other two groups.
Caveat: Small group fMRI data cannot prove individual relapse, craving, or weight-gain risk.