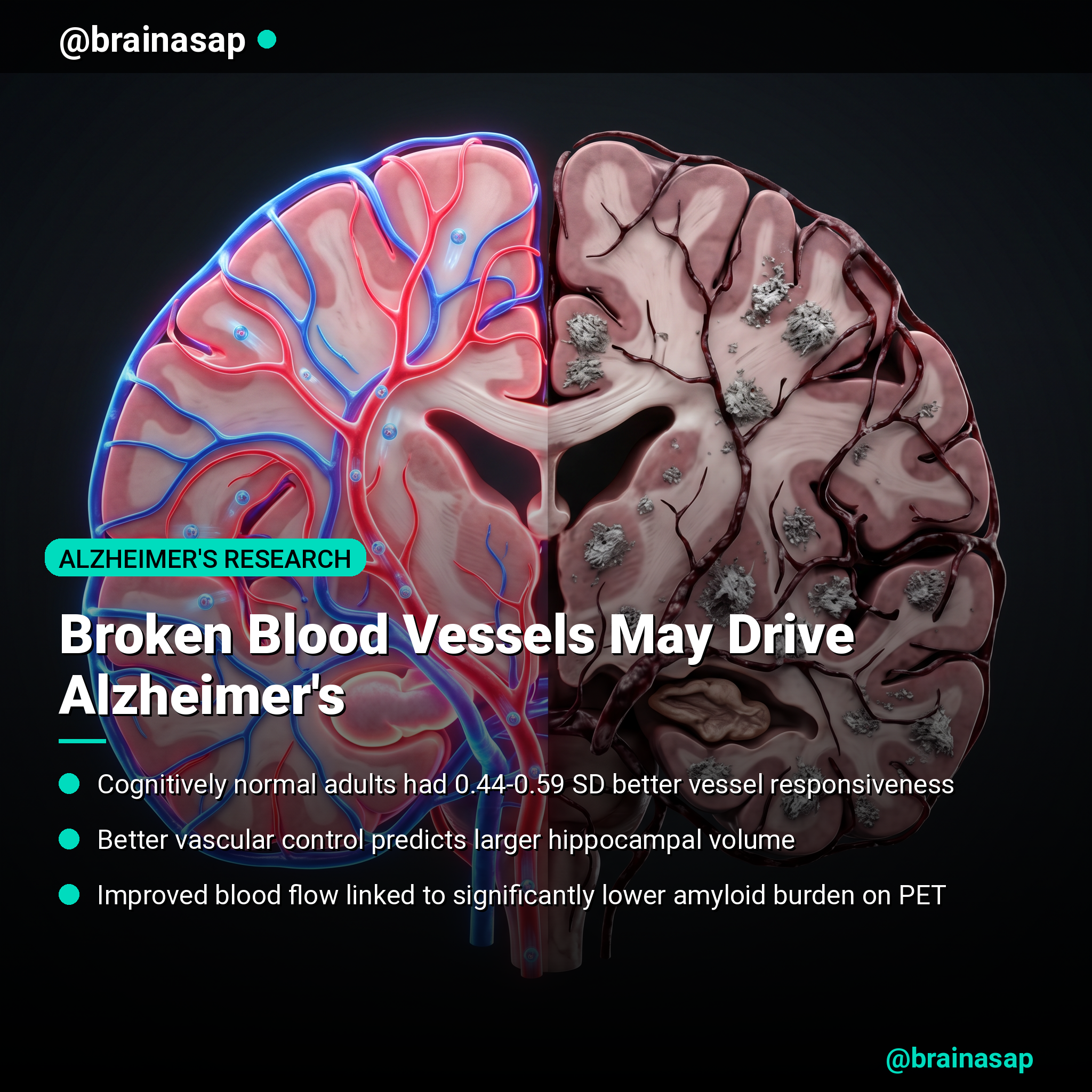
Broken Blood Vessels May Drive Alzheimer’s Decline
TL;DR: A new study reveals that impaired cerebrovascular function—the brain’s ability to regulate blood flow—correlates strongly with Alzheimer’s symptoms, offering a potential non-invasive way to detect early cognitive decline.
Alzheimer’s disease has long been framed as a problem of toxic protein accumulation: amyloid plaques and tau tangles strangling neurons into silence. But what if the disease starts with something more fundamental—a failure of the brain’s plumbing?
Key Findings
- Blood flow dysfunction separates cognitive groups: Cognitively normal adults showed significantly higher hemodynamic indices (measurements of blood vessel responsiveness) compared to those with mild cognitive impairment or dementia—differences of 0.44 to 0.59 standard deviations.
- Vasomotor reactivity predicts brain structure: Higher cerebral oxygenation reactivity (COCR)—a marker of how well the brain’s blood vessels respond to metabolic demands—correlated with greater hippocampal volume, a key region lost in Alzheimer’s disease.
- Better vascular control means less amyloid: Improved dynamic cerebral autoregulation (the brain’s ability to maintain stable blood flow despite pressure changes) was significantly associated with lower global amyloid burden on PET imaging.
- Oxygen delivery matters: Near-infrared spectroscopy revealed that higher cortical tissue oxygenation reactivity correlated with both larger hippocampal volume and reduced amyloid accumulation.
- Non-invasive measurement feasible: These hemodynamic markers were derived from transcranial Doppler ultrasound and near-infrared spectroscopy—inexpensive, portable techniques requiring no participant effort, unlike traditional brain imaging.
- Composite measures show strongest signal: A combined oxygenation reactivity index was significantly associated with lower global amyloid burden, hippocampal volume, and cortical thickness in a temporal Alzheimer’s signature region.
Source: Alzheimer’s & Dementia (2026) | Tsiknia et al.
The Blood Flow Crisis Nobody Talks About
Cerebrovascular dysfunction—the failure of blood vessels to regulate flow and oxygen delivery—may be as important to Alzheimer’s pathology as the plaques we’ve spent decades chasing. Brain imaging studies have long hinted at this connection, but translating vascular dysfunction into clinical markers has been difficult.
This study, led by researchers at USC and collaborating institutions across three sites, took a different approach. Rather than relying on expensive neuroimaging like PET or fMRI, they extracted five distinct hemodynamic measurements from simple ultrasound and light-based spectroscopy—techniques requiring only a few minutes of patient time and costing a fraction of traditional brain scans.
How the Brain’s Blood Vessels Fail
The brain is metabolically voracious: it consumes about 20% of the body’s oxygen despite being only 2% of body weight. Maintaining that supply requires exquisite vascular control. When the arteries and capillaries weaken, oxygen delivery falters, and neuronal metabolism suffers.
The researchers measured five hemodynamic indices derived from dynamic models of blood pressure and CO2 fluctuations. These weren’t static snapshots—they captured how blood vessels respond in real time to metabolic stress. The key markers were:
- Dynamic vascular reactivity (DVR): How quickly cerebral blood flow changes in response to spontaneous fluctuations in arterial pressure.
- Dynamic cerebral autoregulation (DCA): The brain’s ability to maintain constant perfusion despite pressure swings—a critical buffering mechanism.
- Cerebral oxygenation reactivity (COCR): How cortical tissue oxygen levels respond to blood pressure changes.
- Cortical oxygenation pressure reactivity (COPR): How tissue oxygen adapts to end-tidal CO2 fluctuations, measuring metabolic coupling.
A Clear Divide Between Healthy and Impaired Cognition
The findings were striking. Cognitively normal older adults had significantly higher values across all hemodynamic indices compared to those with mild cognitive impairment (MCI) and dementia. The gaps were substantial: for DVR, the difference between normal cognition and dementia was 0.58 standard deviations (p < 0.001).
More importantly, these differences held even after controlling for age, sex, and total cognitive scores—suggesting that vascular dysfunction carries independent clinical weight. A person with similar cognitive test scores to another might still show dramatically different vascular profiles, and those with better vascular function had better structural brain outcomes.
The hippocampus—the seahorse-shaped memory center ravaged early in Alzheimer’s—showed the strongest correlations with hemodynamic indices. Higher COCR predicted larger hippocampal volume (p = 3.7e-03), a correlation that stood after correcting for multiple statistical comparisons. Similarly, better oxygenation reactivity correlated with lower global amyloid burden on PET imaging.
The Bidirectional Blood-Amyloid Connection
Why does better vascular control protect against amyloid accumulation? The mechanisms likely flow both directions. Impaired cerebral blood flow can trigger hypoxia—local oxygen starvation—which may accelerate amyloid production as a metabolic stress response. Conversely, amyloid itself damages blood vessel structure, creating a vicious cycle.
Animal models have shown this loop: when mice overexpress the amyloid precursor protein, their cerebrovascular reactivity declines, reducing oxygen delivery and deepening neuronal hypoxia. That hypoxia then drives further amyloid accumulation. The study doesn’t prove causation in humans, but the correlation—better vascular function associated with lower amyloid burden—suggests this cycle is worth targeting clinically.
The study also found that higher blood pressure variability correlated with worse hemodynamic indices in cognitively impaired adults, even when controlling for mean blood pressure. This hints that chronically dysregulated vascular control may underpin the link between hypertension and dementia risk that epidemiological studies have documented.
Why These Measurements Matter Clinically
MRI and PET imaging provide gold-standard confirmation of brain pathology, but they’re expensive, require specialized infrastructure, and expose patients to radiation (in PET’s case). Transcranial Doppler ultrasound and near-infrared spectroscopy are dramatically cheaper, portable, and can be repeated in a clinic visit without burden.
If these hemodynamic indices prove to be early warning signs of cognitive decline, they could function as screening tools—flagging high-risk older adults for more intensive monitoring or intervention trials targeting vascular health. The study showed that hemodynamic markers differed significantly between cognitively normal and impaired groups, suggesting they might help distinguish subtle cognitive changes that haven’t yet reached diagnostic thresholds.
The real value lies in future longitudinal studies: tracking cognitively normal people with poor vascular function to see if they develop cognitive decline faster. If they do, interventions aimed at vascular repair—whether pharmacological, lifestyle-based, or cardiovascular—could potentially interrupt the Alzheimer’s cascade before amyloid and tau irreversibly scar the brain.
[Insert image: scatter plots comparing hemodynamic indices (DVR, DCA, COCR) across cognitively normal, MCI, and dementia groups]
The Limitations and What Comes Next
The study is cross-sectional—a snapshot in time—which means causation cannot be determined. It’s possible that cognitive decline causes vascular dysfunction, not the reverse. The sample was also relatively homogeneous: mostly non-Hispanic White older adults with low cardiovascular risk, limiting generalization to more diverse populations.
Additionally, only 67 of the 88 cognitively impaired participants had amyloid PET data, slightly reducing statistical power in key analyses. The researchers acknowledge this but note that the associations they found were significant despite the smaller sample, suggesting genuine effects.
The next steps are clear: longitudinal studies tracking how hemodynamic indices predict future cognitive decline in cognitively normal people, and interventional trials testing whether improving vascular function slows or prevents Alzheimer’s progression. The cholinergic and vasculogenic hypotheses of dementia have rarely been tested in tandem—this study suggests they should be.
A New Entry Point for Prevention
For decades, Alzheimer’s research has focused relentlessly on protein misfolding: can we clear amyloid? Can we stabilize tau? Those remain valid targets. But this study joins a growing body of evidence suggesting that the vascular foundation—the physical delivery of oxygen and nutrients—deserves equal attention.
If cerebrovascular dysfunction is indeed a driver of Alzheimer’s pathology, then interventions that improve vascular health—exercise, blood pressure control, cognitive training, or novel vascular-targeted drugs—might show clinical benefit not through dramatic plaques-clearing effects but through the steadier, subtler restoration of brain perfusion. In a disease where early intervention matters, that distinction could be lifesaving.
Citation: Tsiknia AA, Terner JA, Tsokolas ZE, Shin DC, Joe EB, Conti PS, Lepping RJ, Kelley BJ, Zhang R, Billinger SA, Chui HC, Marmarielis VZ, Braskie MN. Cerebrovascular regulation dynamics and Alzheimer’s neuroimaging phenotypes. Alzheimer’s & Dementia. 2026;22:e71146. DOI: 10.1002/alz.71146
Authors’ affiliations: Mark and Mary Stevens Neuroimaging and Informatics Institute, University of Southern California, Los Angeles, CA; Biomedical Engineering Department, USC; Department of Neurology, Keck School of Medicine, USC; Alzheimer’s Disease Research Center, Keck School of Medicine, USC; Molecular Imaging Center, Department of Radiology, USC; Department of Neurology, University of Kansas Medical Center, Kansas City, KS; Department of Neurology, UT Southwestern Medical Center, Dallas, TX; Institute for Exercise and Environmental Medicine, Texas Health Presbyterian Hospital, Dallas, TX.