TL;DR: A new blood test using three misfolded plasma proteins can identify Alzheimer’s disease with 83.44% accuracy, outperforming conventional biomarkers and offering a non-invasive screening tool years before cognitive symptoms appear.
The holy grail of Alzheimer’s research isn’t a cure—yet. It’s catching the disease before memory starts to fade. By the time someone notices confusion, millions of neurons have already died and amyloid plaques have scarred the brain for over a decade. A new study published in Nature Aging reveals that plasma protein misfolding patterns in blood can identify Alzheimer’s status with striking accuracy, potentially transforming how we detect and track the disease.
Key Findings
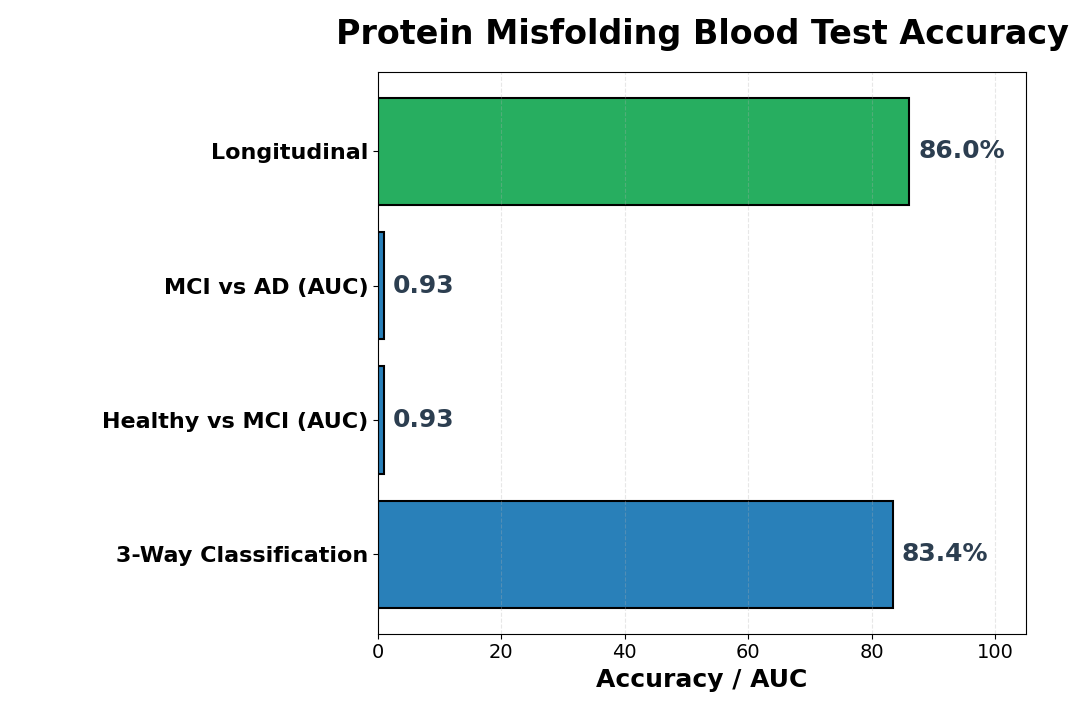
- 83.44% three-way accuracy: A three-protein panel (C1QA, CLUS, ApoB) classified healthy individuals, mild cognitive impairment, and Alzheimer’s disease with 83.44% accuracy across 520 blood samples.
- Superior to standard markers: The protein structural panel outperformed the amyloid-beta/tau ratio (AUC 0.934 vs 0.742), the conventional blood biomarker for AD diagnosis.
- High sensitivity across disease stages: The test detected healthy versus mild cognitive impairment with AUC 0.9325, and mild cognitive impairment versus Alzheimer’s with AUC 0.9343, indicating reliable discrimination across the disease spectrum.
- Longitudinal stability: In follow-up samples spanning 255 days, the three-protein panel maintained 86% accuracy at tracking disease progression, even as individual protein signatures shifted.
- Age and sex variations identified: Structural protein changes showed distinct neuropsychiatric patterns in males and females, with women exhibiting more rapid cognitive decline and ApoE ε4 carriers showing heightened vulnerability.
- APOE genotype modulates risk: Among APOE e4 carriers, the accessibility of misfolded proteins increased significantly, suggesting genetic background amplifies pathological protein conformations.
Source: Nature Aging (2026) | Son et al.
How Protein Misfolding Reveals Hidden Alzheimer’s
Most Alzheimer’s biomarkers chase the same two suspects: amyloid-beta and tau, the hallmark proteins that clump in the brain. But this study took a different approach. Researchers focused on structural changes in plasma proteins—the way proteins fold, misfold, and interact—rather than abundance alone. The reasoning is elegant: if Alzheimer’s disrupts brain proteostasis (the system that keeps proteins properly folded), those changes should ripple outward into the bloodstream.
The team used a technique called cross-linked mass spectrometry to map protein structures at atomic resolution, then applied machine learning to identify which conformational signatures separated healthy brains from diseased ones. What emerged was striking: three proteins—C1QA (complement), CLUS (clusterin), and ApoB (apolipoprotein B)—showed consistent structural abnormalities across AD cases, and these weren’t just noise. They were robust enough to build a diagnostic algorithm.
The Three-Protein Panel: A Practical Blood Test
Why these three? C1QA and clusterin are ancient immune and protein-folding proteins with documented links to amyloid clearance and neuroinflammation. ApoB is a structural protein central to cholesterol metabolism and blood-brain barrier function—and mounting evidence implicates it in AD pathogenesis. The researchers hypothesized that the structural accessibility of these proteins (how exposed or buried their amino acid residues were) would shift when proteostasis breaks down in AD.
They were right. In the 520-person discovery cohort (healthy, mild cognitive impairment, Alzheimer’s), the panel achieved 83.44% three-way accuracy. More impressively, it delivered AUC 0.9325 for healthy versus MCI and AUC 0.9343 for MCI versus AD—margins that rival or exceed existing plasma biomarkers like amyloid-beta/tau ratios (AUC 0.742). The practical payoff: a simple blood draw could screen asymptomatic individuals years before cognitive decline becomes apparent, enabling preventive intervention at the optimal window.
Protein Misfolding as a Window Into Neuropsychiatric Burden
Beyond diagnosis, the study uncovered something unexpected: the accessibility of these three proteins correlated strongly with specific neuropsychiatric symptoms. Depressed mood, anxiety, apathy, and sleep disturbance—hallmark features of AD—each showed distinct relationships to protein structural changes. Female participants with AD exhibited more rapid cognitive impairment, and this sex difference mirrored their protein accessibility profiles, hinting that males and females may experience different proteomic perturbations as the disease advances.
The APOE genotype—the strongest genetic risk factor for late-onset AD—modulated these effects. Individuals carrying the e4 allele showed heightened accessibility changes across all three proteins, suggesting that genetic susceptibility amplifies proteostasis dysfunction. This opens a testable hypothesis: APOE e4 carriers may benefit from earlier screening and more aggressive intervention, a personalized medicine angle that warrants investigation.
Longitudinal Validation: Does It Track Disease Progression?
A blood test is only useful if it remains accurate over time and tracks meaningful disease changes. The team followed 50 individuals (25 healthy, 12 with AD, others with MCI) over 255 days. The three-protein panel maintained 86% accuracy at classifying disease stage across follow-up visits, even as individual peptide accessibility shifted. This stability suggests the panel captures a robust underlying pathophysiology rather than noise.
Intriguingly, longitudinal changes were asymmetric: some individuals transitioned from healthy to MCI or MCI to AD, while others remained stable despite minor accessibility fluctuations. This hints that the panel might eventually help predict who will progress and who won’t—a critical capability for clinical trials seeking enriched populations or for counseling patients on prognosis.
What About Confounders and False Alarms?
The researchers validated their approach against two independent cohorts (UCSD and KUMC) and tested robustness to known confounders: MMSE and CDRSUM cognitive scores, age, sex, vascular risk factors, and depression severity. The panel held up. Age-stratified performance showed consistent accuracy in older adults (over 70) without drift, though younger individuals showed slightly lower discriminatory power—probably because early AD is rarer in this age group.
One caveat: the panel’s accessibility scores didn’t explain all disease variability. Some healthy individuals carried accessibility signatures resembling MCI or AD, while some MCI patients resembled healthy controls. This residual heterogeneity likely reflects the biological complexity of early AD and the possibility that some individuals have silent pathology yet to manifest as symptoms. Ongoing longitudinal follow-up of misclassified individuals may reveal whether the test predicts future cognitive decline in those currently asymptomatic.
Clinical and Research Implications
If validated in prospective studies, this three-protein panel could reshape AD screening. Current guidelines recommend PET imaging or CSF tau measurements for amyloid pathology confirmation—expensive, invasive, or both. A blood test with 83% accuracy would democratize early detection, making screening accessible in primary care settings where most older adults are seen. It could identify candidates for anti-amyloid monoclonal antibodies (aducanumab, lecanemab) at the preclinical stage when these drugs show the greatest cognitive benefit.
For research, the panel offers a faster, cheaper way to stratify participants in prevention trials and track therapeutic response. The sex- and genotype-specific patterns hint that future algorithms might personalize risk prediction: an APOE e4 carrier might warrant more frequent screening or lower treatment thresholds than an e3 homozygote.
The Bigger Picture: From Protein Folding to Prevention
This work underscores a shift in Alzheimer’s biology: from counting amyloid plaques to understanding proteostasis—the cellular systems that maintain protein quality and prevent misfolding. As proteostasis declines with age and genetic risk, proteins throughout the body (not just in the brain) begin to fray and interact abnormally. Measuring these structural changes in plasma may be a better proxy for brain pathology than looking for amyloid or tau alone, which can persist asymptomatically for years.
The three-protein signature—C1QA, clusterin, and ApoB—likely reflects distinct biological mechanisms: immune dysregulation, impaired protein chaperone activity, and lipid metabolism disruption. By identifying these specific proteins, researchers have created a window into the molecular chaos of early Alzheimer’s that transcends anatomy. A blood test based on this principle could one day be as routine as a cholesterol screening, catching the disease before symptoms steal memory.
Citation: Son O, et al. Structural signature of plasma proteins classifies the status of Alzheimer’s disease. Nature Aging. 2026;6:597–611. DOI: 10.1038/s43587-026-01078-2
Authors’ affiliations: Department of Molecular Medicine, The Scripps Research Institute, La Jolla, CA; Department of Medical Science Convergence, Graduate School of Medical Science, University of Ulsan, Republic of Korea; Department of Convergent Bioscience and Informatics, Chungnam National University; University of Kansas Alzheimer’s Disease Research Center; University of Southern California; Alzheimer’s Therapeutic Institute.