TL;DR: Octogenarians who recall words like 50-year-olds carry a distinct brain profile: preserved cortical volume, a cingulate cortex thicker than younger adults, larger entorhinal neurons, fewer inflammatory microglia, and more von Economo neurons — the biology is real, not just motivational.
Key Findings
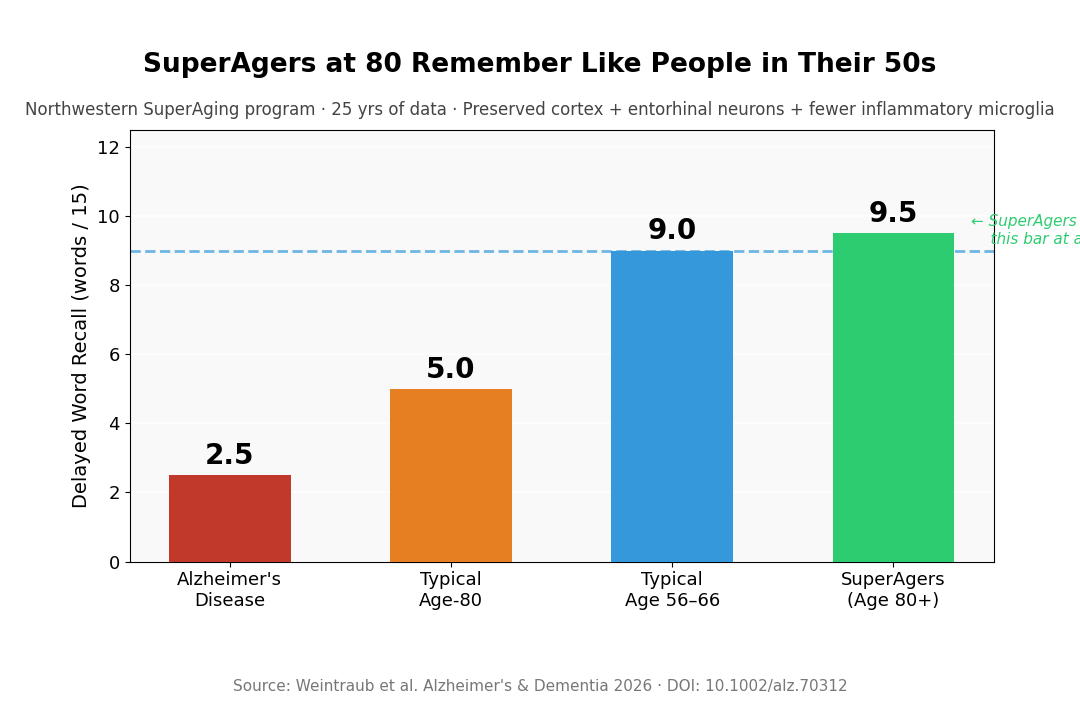
- Age-80 memory matched 50-to-60-year-olds: SuperAgers are defined as adults ≥80 whose delayed word recall equals or exceeds performance typical of people 20–30 years younger.
- “Normal” aging hides a lot of loss: Typical delayed recall at 80 averages ~5/15 words versus ~9/15 at ages 56–66 — average isn’t intact, it’s just familiar decline.
- Cortical volumes looked decades younger: SuperAger brains showed cortical volumes comparable to adults 20–30 years their junior rather than age peers.
- Cingulate cortex was thicker than in younger adults: One cingulate region was unusually preserved — not just spared, but exceeding typical younger-adult thickness.
- Postmortem tissue showed cellular resilience across multiple systems: Fewer Alzheimer-type changes, larger Layer II entorhinal neurons, fewer inflammatory microglia in white matter, better-preserved cholinergic innervation, and more von Economo neurons.
- Perspective review of 25-year research program: This is a synthesis paper from Northwestern’s SuperAging cohort — not a new clinical trial, but a consolidated account of what the biology of exceptional memory aging looks like.
Source: Alzheimer’s & Dementia (2026) | Weintraub et al.
The usual frame for late-life cognition is defensive: how to slow decline, how to delay diagnosis, how to reduce risk. Northwestern’s SuperAging program inverts the question. Instead of asking why some people lose memory as they age, it asks why some people don’t — and what their brains are doing differently.
SuperAgers are defined stringently: adults 80 or older whose delayed word recall meets or exceeds the performance of people 20–30 years younger. That isn’t just “aging well.” It’s maintaining a cognitive trajectory that most people in their 50s and 60s can’t match.
After 25 years of studying this population, the Northwestern group has accumulated evidence that exceptional memory preservation in old age is a biological phenomenon, not a lifestyle brand.
SuperAgers vs. Normal Aging
The paper makes a point that’s easy to miss but quietly reframes everything else. Average delayed word recall at age 80 is around 5 out of 15 words. At ages 56–66, it’s closer to 9 out of 15. That difference — nearly halved — is what typically gets called “normal cognitive aging.”
SuperAgers exceed the 56-to-66-year-old threshold at age 80 or beyond. The benchmark isn’t cherry-picked; it’s based on normative performance data from younger adults. What it exposes is that “normal” late-life cognition is already substantially declined relative to midlife — it’s just declined across the board, so it gets treated as the floor rather than a loss.
That reframing matters for how we think about dementia prevention. If average 80-year-old cognition is already at 55% of midlife performance, then preventing Alzheimer’s isn’t the only goal. Understanding why some people arrive at 80 with midlife memory intact is a separate and equally valid target.
The Cortex Looked Decades Younger
The structural MRI findings from the Northwestern cohort are consistent and striking. SuperAger cortical volumes resemble those of adults 20–30 years younger — not just relative to age-matched peers, but in absolute terms. The typical age-related cortical thinning that accumulates through the 70s and 80s is largely absent.
The cingulate finding goes further. One cingulate region was thicker than in younger neurotypical adults — not just preserved but anomalously robust. The cingulate cortex is involved in attention, salience, and higher-order memory processes; a region that’s thicker in 80-year-olds than in 50-year-olds isn’t just avoiding decay, it may be reflecting an unusually durable or developmentally distinctive network organization.
The Cellular Findings Hit Alzheimer’s Vulnerable Systems
The postmortem brain tissue data are where the story becomes most compelling. SuperAgers show multiple tissue-level differences from typical older adults:
Entorhinal neurons in cortical Layer II are unusually large. The entorhinal cortex is one of the first regions targeted by Alzheimer’s pathology — this is where the tau tangles of early disease typically begin spreading. Larger, healthier neurons in this region are a direct indicator of preserved architecture in a system that usually fails first.
Inflammatory microglia are fewer in SuperAger white matter. Microglia are the brain’s resident immune cells; when chronically activated, they contribute to neuroinflammation and accelerate neurodegenerative processes. A less inflammatory white matter environment fits a broader pattern of reduced pathological aging.
Cholinergic innervation is better preserved. The cholinergic system — the same one depleted in Alzheimer’s and targeted by drugs like donepezil — supports memory consolidation and attentional modulation. SuperAgers retain more of it.
Von Economo neurons are more abundant. These large, spindle-shaped cells are concentrated in regions involved in social cognition, emotional salience, and self-awareness, and are preferentially lost in several neurodegenerative conditions. Their higher density in SuperAgers may reflect preserved social-processing networks — which aligns with behavioral observations that many SuperAgers are highly socially engaged.
This Is a Resilience Phenotype, Not a Single Magic Region
The pattern across all these findings is not that SuperAgers have one special brain structure or one protected mechanism. It’s that they preserve multiple interdependent systems simultaneously: cortical architecture, cholinergic input, inflammatory tone, vulnerable memory circuit neurons, and specialized cell populations linked to social and emotional cognition.
Memory isn’t a single function stored in a single region. It depends on distributed networks — medial temporal circuits for formation, prefrontal systems for working memory, attention networks for encoding, cholinergic input for gating and plasticity. SuperAgers appear to keep several components of that system at something closer to midlife performance than any individual factor would predict.
That’s what makes the SuperAger phenotype scientifically valuable: it identifies a positive-control group for resilience research. Instead of only comparing Alzheimer’s disease to average aging, researchers can compare both to exceptional aging — and ask which of the aging changes between those states are unavoidable biology versus modifiable or delayed processes.
What SuperAging Research Can’t Yet Answer
The temptation is to immediately turn the SuperAger profile into a lifestyle checklist: stay socially engaged, exercise, sleep well, keep learning, manage cardiovascular risk. Those habits are reasonable recommendations regardless.
But the biology of SuperAging almost certainly involves genetic factors, early-life development, and individual variation in cellular resilience that can’t be fully created in late life by behavioral change.
The more productive scientific questions are: Which SuperAger features are causes of resilience, which are consequences of preserved brain health, and which are markers of a third underlying variable? Does social engagement drive von Economo neuron preservation, or do both reflect a shared underlying neurological trait? Can any biomarker identify likely SuperAgers in midlife, before the outcomes are visible?
The field’s next challenge is connecting the cellular postmortem findings to in-vivo blood biomarkers and imaging that can track the same individuals longitudinally. If SuperAgers can be identified before age 80 — by biomarker profile, cognitive trajectory, or genetic signature — the window for understanding and potentially extending their resilience opens considerably.
The bottom line from 25 years of this work: late-life memory preservation at a level that embarrasses average 80-year-olds is not just luck or a good attitude. It has anatomy, cellular structure, and system-level biology behind it. That biology is now worth taking apart systematically.
Citation: Weintraub et al. Superaging and resistance to involutional cognitive decline. Alzheimer’s & Dementia. 2026. DOI: 10.1002/alz.70312
Study Design: Perspective review of the Northwestern SuperAging research program (25 years of cohort data, neuroimaging, and postmortem neuropathology).
Sample Size: SuperAgers defined as adults ≥80 with delayed word recall ≥ normative performance of 56-to-66-year-olds.
Key Statistic: Typical delayed recall at 80: ~5/15. Typical at 56–66: ~9/15. SuperAgers meet or exceed the younger benchmark. Postmortem: fewer Alzheimer changes, larger entorhinal neurons, less microglia inflammation, more von Economo neurons.