TL;DR: A 2026 analysis in European Heart Journal found that a lower vigorous-activity share was more strongly tied to dementia-attributable risk than lower total activity volume in UK Biobank accelerometer data.
Key Findings
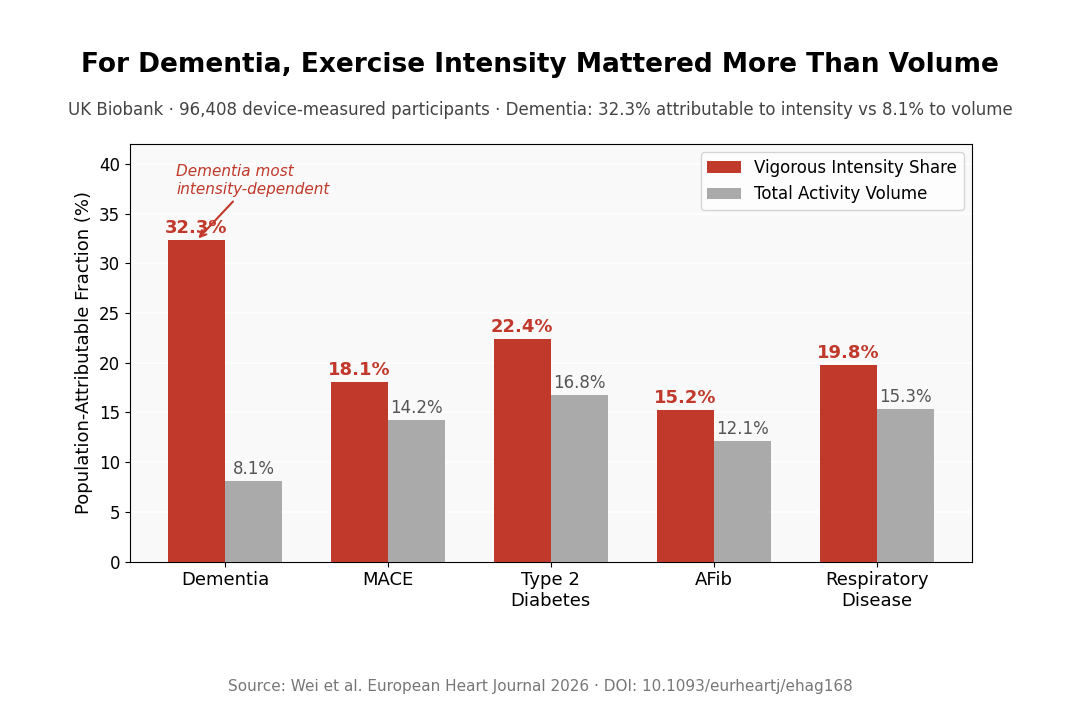
- Dementia favored intensity over volume: Population-attributable analysis estimated a larger dementia burden tied to low vigorous-intensity share than to low total activity volume — 32.3% versus 8.1% in the model.
- >4% vigorous activity linked to 29–61% lower risk across eight diseases: Compared to 0% vigorous physical activity (VPA), participants with more than 4% VPA had substantially lower adjusted risks for MACE, type 2 diabetes, AFib, dementia, and others.
- Intensity effect held after controlling for total volume: The vigorous-activity advantage appeared in analyses that already accounted for total movement — intensity retained independent risk information.
- Accelerometers captured what self-report often misses: Device-measured wrist data detected short vigorous bursts — brief stair climbs, fast uphills, carrying loads — that questionnaires typically miss.
- UK Biobank scale: 96,408 device-measured + 375,730 self-reported participants: Eight chronic disease outcomes plus all-cause mortality tested across both datasets.
- Observational — healthy-user bias can’t be fully removed: Healthier people can perform more vigorous exercise, so residual confounding remains even with adjustment.
Source: European Heart Journal (2026) | Wei et al.
Most physical-activity research separates people by total movement volume. This UK Biobank analysis tested whether the vigorous share of daily movement predicted dementia risk beyond total activity volume, then compared that finding with cardiovascular disease, cancer, and all-cause mortality outcomes.
Accelerometers to Track Exercise in UK Biobank Data
Self-reported exercise has a known measurement problem: people remember formal workouts better than embedded physical activity, and intensity categories are imprecise. Accelerometer data let researchers separate total movement from the fraction performed at higher intensity.
Wrist accelerometers fixed this. They captured movement continuously, including the short, intense bursts that often don’t get logged — hurrying upstairs, carrying groceries quickly, walking fast to catch a bus.
Device data let researchers separate total physical activity from the percentage of that activity classified as vigorous, then test whether that vigorous fraction predicted disease risk independently of total volume.
Across 96,408 participants with device data, the vigorous share still predicted outcomes after accounting for total activity. Two people with the same step count could fall into different risk groups if one regularly included brief hard efforts and the other did not.
Dementia Stood Out Among Eight Chronic Diseases
The study tested 8 chronic disease categories plus all-cause mortality: major cardiovascular events, atrial fibrillation, type 2 diabetes, inflammatory disease, metabolic liver disease, respiratory disease, kidney disease, and dementia.
- Intensity: lower vigorous share of daily movement was the exposure most tied to higher dementia risk.
- Volume: total movement still mattered, but low total volume explained less of the model-estimated dementia burden.
- Interpretation: short breathless activity bursts should be tested separately from step counts, because the association is not just a total-movement signal.
Vigorous activity was associated with lower risk across outcomes, but dementia was the most intensity-dependent result. In the population-attributable model, low vigorous-intensity share accounted for an estimated 32.3% of dementia burden, compared with 8.1% for low total activity volume.
That wording matters: the model treats little or no vigorous-intensity movement as the risk-side exposure. People with less vigorous-intensity movement carried more of the modeled dementia burden, even when total movement volume was considered.
For every other disease in the study, the intensity-volume pattern looked different.
That asymmetry suggests the brain may respond differently to movement intensity than to duration alone. One possible explanation is vascular reserve: harder effort places stronger demands on blood-flow regulation, and cerebrovascular health is central to dementia risk.
Other possible explanations include metabolic flexibility, inflammatory control, neurotrophic signaling, or some combination of pathways that higher-intensity movement may challenge more than sustained gentle movement does.
Vigorous Activity Threshold Started Near 4% of Daily Movement
The headline comparison — participants with more than 4% vigorous physical activity versus those with 0% — showed 29–61% lower risks across disease categories. That 4% threshold sounds abstract, but it isn’t.
In an average waking day’s movement, 4% vigorous means perhaps a few minutes of genuine breathlessness: a fast uphill, a heavy carry, a stair sprint.
This doesn’t require an hour at the gym. It requires some moments of real effort embedded in daily life.
The study doesn’t prove that adding those bursts is what causes the lower risk — it’s observational, and healthier people are more capable of vigorous effort. But the finding that even modest vigorous fractions separated risk groups is a clinically useful clue for public-health framing.
Vigorous Activity Was More Dementia-Relevant Than Daily Step Counts
The dementia-intensity pattern fits what’s known about the mechanisms linking exercise to brain aging. Vascular health is one of the strongest modifiable determinants of dementia risk — and vigorous exercise challenges the cardiovascular system in ways that low-intensity sustained movement does less effectively.
Dementia also shares risk factors with metabolic and inflammatory disease. Insulin resistance, blood pressure variability, chronic inflammation, and cardiorespiratory fitness all shape the brain’s long-term environment.
Vigorous exercise may address several of these simultaneously: improving endothelial function, reducing arterial stiffness, enhancing insulin sensitivity, and driving anti-inflammatory adaptations that moderate-intensity activity produces less efficiently.
There may also be a neurotrophic component. BDNF — brain-derived neurotrophic factor, a protein that supports neuron survival and plasticity — has been shown to rise more robustly with higher-intensity exercise in several studies.
Whether that acute effect translates into long-term dementia protection is unresolved, but it’s a plausible pathway.
UK Biobank Accelerometer Data Do Not Prove Sprinting Prevents Dementia
The observational design leaves the conclusion appropriately conditional. Healthier people exercise harder, and no statistical model fully removes that reverse-causality problem.
Some of the dementia-vigorous-exercise association may reflect brain health enabling vigorous movement, not vigorous movement protecting the brain.
There’s also the question of who this advice applies to. For people with cardiovascular disease, musculoskeletal problems, or unstable chronic illness, “add some vigorous bursts” needs to be medically supervised and individually calibrated.
This study doesn’t override that context.
What it does provide is a better target for exercise research and public-health messaging. The current default — move more, sit less, aim for 150 minutes of moderate activity per week — is correct but incomplete.
For dementia specifically, the intensity of movement appears to carry independent weight that can’t be accumulated through gentle strolling alone.
The clearest next test would be a randomized trial that holds total activity volume constant while varying the vigorous fraction — tracking cognition, cerebrovascular markers, metabolic health, and inflammatory biomarkers as outcomes.
Until then, the UK Biobank finding gives the field well-powered, device-measured evidence that intensity deserves its own line in exercise prescriptions for brain aging.
Citation: DOI: 10.1093/eurheartj/ehag168; Wei et al; Volume vs intensity of physical activity and risk of cardiovascular and non-cardiovascular chronic diseases; European Heart Journal; 2026.
Study Design: Prospective UK Biobank cohort; wrist accelerometer data (n=96,408) and IPAQ self-reported activity (n=375,730).
Sample Size: 96,408 (device-measured); 375,730 (self-reported).
Key Statistic: Dementia population-attributable fraction: 32.3% for low vigorous-intensity share vs 8.1% for low total activity volume. >4% VPA was linked to 29–61% lower adjusted risks across 8 chronic diseases vs 0% VPA.