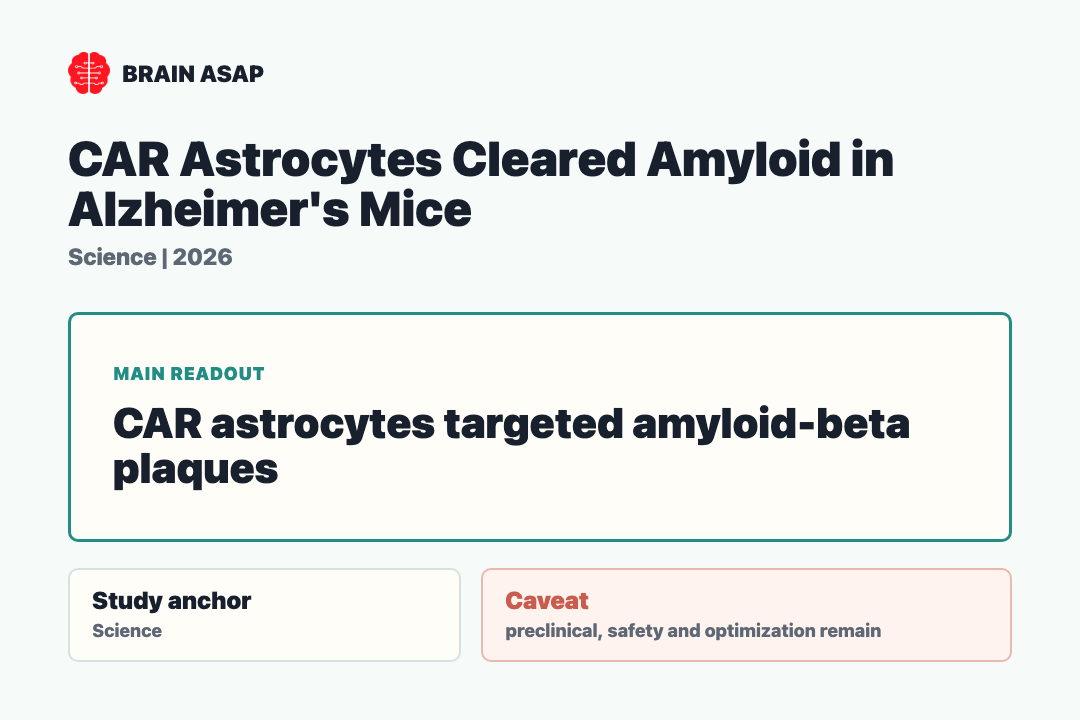
TL;DR: A 2026 mouse study in Science engineered CAR astrocytes to target amyloid-beta, clearing plaques in young Alzheimer’s-model mice and reducing existing plaque burden in older mice.
Key Findings
- Plaque-free at 6 months in the prevention arm: Young Alzheimer-model mice treated before plaques formed remained plaque-free at the age when untreated mice are normally saturated.
- ~50% reduction in established plaques: Older mice with existing amyloid pathology had roughly half the plaque burden of control-virus mice after one CAR-A treatment.
- Astrocytes got a target via viral gene delivery: A chimeric antigen receptor designed to recognize amyloid-beta was installed in resident brain support cells.
- Coordinated glial response, not just astrocytes: Single-nucleus RNA sequencing showed astrocytes and microglia shifting together — the local glial ecosystem reorganized around amyloid.
- Single-treatment durability is the differentiator from antibodies: Lecanemab and donanemab require repeated high-dose infusions; CAR-A aims for a durable cellular cleanup system after one delivery.
- Mouse data, not clinic-ready: Researchers explicitly flag optimization, safety, and side-effect work before any human translation.
Source: Science (2026) | Chen et al.
CAR therapy usually means engineered immune cells hunting cancer. CAR-astrocyte therapy tests a stranger idea: what if the brain’s own support cells could be given a target and a job?
Instead of dosing an antibody from outside the brain every 2 weeks, give the glia already living next to amyloid plaques the receptor and tools to clean them up themselves.
In the mouse experiments, engineered astrocytes recognized amyloid-beta plaques and reduced plaque burden.
Engineered CAR Astrocytes Targeted Amyloid-Beta Plaques
Astrocytes are usually described as support cells, but the phrase undersells them. They maintain the brain’s chemical environment, respond to injury, regulate the blood-brain barrier, and signal back and forth with microglia.
They are also already inside the tissue that monoclonal antibodies struggle to reach in adequate quantities.
The WashU team’s design borrows the CAR-T template and applies it to glia. A viral construct delivers a chimeric antigen receptor tuned to recognize amyloid-beta.
The engineered astrocytes can then identify plaques and trigger removal — without crossing the blood-brain barrier from the bloodstream every time, because they are already on the right side of it.
Two design choices make or break the strategy. The viral construct has to reach enough astrocytes and express the receptor strongly enough to matter, without pushing the cells out of their normal support roles.
And target specificity has to be tight enough that the engineered receptor binds plaques without misreading normal proteins, vessels, or glial signaling partners.
One CAR Astrocyte Injection Reduced Amyloid in Two Mouse Tests
The team tested CAR-A in two scenarios that map onto how Alzheimer’s actually arrives clinically. In the prevention arm, young Alzheimer-model mice received the construct before plaques developed.
In the cleanup arm, older mice received it after the brain was already plaque-saturated.
The prevention result was the cleanest. Treated young mice were described as plaque-free at nearly 6 months — the age when untreated Alzheimer-model mice are normally heavily plaque-loaded.
In the treatment arm, existing amyloid plaque burden fell by about 50% compared with control-virus animals.
One injection. Two scenarios. Two clear results.
Mouse Microglia Remained Present After CAR Astrocyte Treatment
One concern when engineering one glial population is that you might just be substituting it for another — quietly disrupting the normal division of labor between astrocytes and microglia. That is not what happened.
Single-nucleus RNA sequencing showed a coordinated glial response: astrocytes and microglia shifted together around the amyloid pathology.
that ecosystem-level response changes interpretation. Alzheimer’s is not just a plaque-storage problem.
The brain’s immune and support cells respond to plaques in different ways. A therapy that rewires astrocytes can also change nearby microglia, inflammatory signaling, and synaptic tissue state, so the safety question is whether plaque clearance comes with harmful glial activation.
Plaque count alone cannot answer that. Cell-state sequencing let researchers look beyond the headline number.
Antibody Comparison Tested Whether Plaque Targeting Was Specific
Researchers contrasted CAR-A with monoclonal antibodies like lecanemab and donanemab. The comparison is honest about what each evidence base supports.
Antibodies have human outcome data and known safety problems — ARIA, infusion logistics, modest cognitive effect sizes. CAR-A has a striking mouse plaque result and a long list of unanswered questions about control, reversibility, and long-term glial behavior.
The structural difference is durability. Repeated antibody infusion and one-time cellular engineering create different risk profiles.
Antibodies can be stopped if problems appear. A persistent engineered glial program would need built-in safety controls or evidence that expression fades predictably.
Long persistence could make a single treatment more powerful — and also raises the bar for proving controllability before human testing.
CAR Astrocytes Add a Plaque-Targeting Cell-Therapy Strategy
The important result is not just that plaques fell. Astrocytes, usually treated as background biology in amyloid stories, became programmable therapeutic actors.
If the strategy can be made safe, targeted, and durable, it would expand Alzheimer’s therapy beyond antibodies and into engineered brain-resident cells.
The safety questions are different from CAR-T. Astrocytes regulate synapses, extracellular ions, blood-brain barrier interactions, and metabolic support — engineering them has to preserve normal housekeeping while adding amyloid recognition.
That is a more constrained problem than getting an immune cell to attack tumors.
Mouse CAR Astrocyte Results Still Need Human Safety Testing
Evidence so far comes from mice, not patients. Young Alzheimer-model mice were treated before plaques formed; older plaque-saturated mice were treated after pathology was established.
A result can be scientifically valuable inside that boundary and easy to overextend outside it.
Three honest framings to keep in mind:
- Amyloid burden is not cognition. A clean plaque result in mice does not equal memory benefit in humans — the field already learned this lesson with antibodies.
- Alzheimer’s is more than amyloid. Tau pathology, synapse loss, vascular injury, and immune state all shape whether plaque clearance translates to clinical change.
- Durability cuts both ways. A persistent therapeutic program is powerful and harder to reverse. Safety design has to start from that asymmetry.
A stronger follow-up would measure cognition, synaptic integrity, gliosis, and tau-linked biology alongside plaque load. If CAR-A only clears amyloid without preserving function, it stays a powerful mechanistic tool rather than a disease-modifying therapy.
The broader platform problem is real, though.
If astrocytes can be safely programmed in the brain, the same logic could eventually be tested against other extracellular targets — but only after the amyloid model proves engineered glia can be controlled over time.
The useful framing is not “antibodies are obsolete.” It is that brain-resident cells can be enrolled as durable therapeutic agents, a treatment strategy that has not been available in Alzheimer’s disease.
Citation: DOI: 10.1126/science.ads3972; Chen et al; Targeting amyloid-beta pathology by chimeric antigen receptor astrocyte (CAR-A) therapy; Science; 2026.
Study Design: Preclinical CAR-astrocyte engineering study with in vitro validation, Alzheimer mouse prevention and treatment experiments, and single-nucleus RNA sequencing.
Sample: Young Alzheimer-model mice treated before plaques formed; older plaque-saturated mice treated after pathology was established.
Key Result: Single CAR-A treatment kept young mice plaque-free at ~6 months and reduced existing amyloid plaque burden by ~50% in older mice.
Caveat: Amyloid clearance in mice is not cognitive benefit in humans; safety, controllability, and durability still need to be established.